HTIRC E-NEWSLETTER
Fall and Winter 2018
Volume 11 Issue 2
Articles in this issue
- HTIRC Butternut Breeding and Research Program
- Stratification and Germination of Pure Butternut (Juglans cinerea) and Hybrid Butternut (Juglans x bixbyi) Seeds
- Tax Deductions for Casualty Loss of Timber
- An Update on Chestnut Blight Research at the HTIRC
- Thousand Cankers Disease Best Management Practices Recommendations
- Thousand Cankers Disease: An Indiana Perspective
- Applications of Pest Monitoring Data in Intensively Managed Walnut Plantations
- HTIRC Partner Highlighting Their Work on Facebook
- HTIRC Tree Improvement 2018 Seed Harvest and Distribution of Seed
- New HTIRC Research Grants to Advance Strategic Plan Goals
- Conference Announcements
- Honors and Graduations
- 2018 Research Publications
Welcome to this issue of the Hardwood Tree Improvement and Regeneration Center E-newsletter. We have a new website and associated newsletter format that we hope you will enjoy and find useful.
The HTIRC is committed to enhancing the productivity and quality of Central Hardwood Region trees and forests for the economic and environmental benefits they provide. Scientists at the HTIRC are using conventional tree improvement breeding as well as molecular and genetic technologies to improve the wood quality, growth characteristics, and insect and disease resistance of trees like black walnut, black cherry, red and white oaks, butternut and American chestnut. Research in tree breeding, tree nursery practices, tree plantation establishment and management, and Central Hardwoods silvicultural systems is aimed at increasing the regeneration success rate for high quality hardwood trees and forests.
We invite you to read about some of our activities by visiting the E-Newsletter Page at: https://htirc.org//resources/newsletters/
If you wish to download the pdf version and your web browser is Internet Explorer, we recommend you first download Adobe Acrobat Reader DC at https://get.adobe.com/reader/ for the newsletter format to appear correctly on your screen.
HTIRC Butternut Breeding and Research Program
by Jim McKenna, Operational Tree Breeder
Butternut, Juglans cinerea, also known as white walnut, has been a target of HTIRC research for over a decade. Butternut, although never common, is now a rare tree across most of its former range due in part to a fungal pathogen called butternut canker disease. Our research, done in cooperation with many partners, has concentrated on collecting and screening native butternut and naturally occurring hybrids between butternut and Japanese walnut for disease resistance. We hoped to identify disease resistance in native butternut, but also investigated the hybrids in case disease resistance in native butternut populations was weak of absent. Disease-resistant or tolerant families could be propagated for butternut restoration plantings on public and private lands to restore this tree species with historic importance for wildlife food and high value timber products.
Findings, as of 2018, from our research program are summarized below:
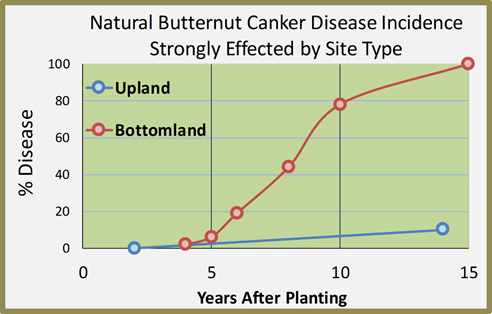
- Determined the highly significant suppression of butternut canker disease on upland sites vs. bottom land sites. Monitored plantings on upland sites have seen much slower progression of natural disease spread than on bottomland sites. This observation led to the initiation of a “citizen science” project supplying butternut seedlings to landowners and public land managers in Ohio and Indiana for paired plantings on upland and bottomland sites to investigate further our initial observations. Reports from these plantings will help us evaluate this disease response difference over a much wider range of sites.
- Determined that screening seedlings naturally underneath diseased over-story trees was much faster (4 years compared to 10-years) and more economical and informative than previous inoculation methods. Previously, individual trees were inoculated with fungal cultures requiring significant time and effort to screen for potential resistance. We discovered that retaining a few infected but surviving trees in previous plantings and replanting new seedlings adjacent to those retained trees results in more rapid and efficient inoculation of the seedlings and reflects a more natural means of infection.
- Evaluated the 2nd screening block of HTIRC-Purdue (10-years post inoculation) and found hybrid families possess much more resistance than do pure butternut families. We have found butternut canker disease resistance is weak or absent in the families of butternut we have screened. Some families of hybrid butternut demonstrate much stronger resistance. The families of butternut and hybrid butternuts demonstrating the best resistance provide potential candidates for breeding and seed orchard trees to move toward butternut restoration planting stock. Note that the seed family 15-86 is a new seed accession from New York State that was called a pure J. cinerea but its disease reaction and twig morphology suggest hybrid.
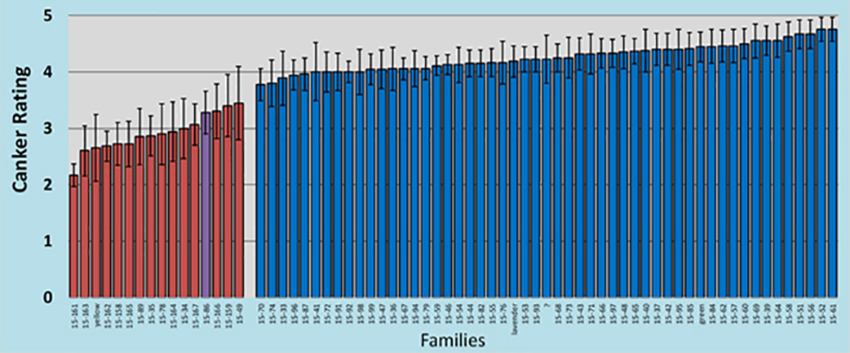
Red bars represent hybrid butternut (hybrids with Japanese walnut) and blue bars represent pure butternut. Canker ratings are based on the number of cankers per tree with “1” being none to very few and “5” being many.

We have reached an agreement to begin to supply excess pure butternut seed from our Ohio and Indiana orchards to the Clements State Tree Nursery in West Virginia, of the West Virginia Division of Forestry, to grow and provide landowners a future supply of butternut. 2018 marks the 10th year the IN-DNR Division of Forestry has been providing hybrid butternut seedlings to Indiana residents through their state forest nursery system. The IN-DNR has been a key cooperator utilizing our screening data and the first state in the native range to deploy hybrid butternut. Beside data, we have shared excess hybrid seed and selections for new hybrid orchards.
The cooperation and contributions of many agencies, landowners, resource professionals and researchers has made this restoration project possible by providing butternut seed or seedlings, planting sites, seedling production, and distribution assistance.
Stratification and Germination of Pure Butternut (Juglans cinerea) and Hybrid Butternut (Juglans x bixbyi) Seeds
by Andrea Brennan and Douglass Jacobs
Part 1: Seed Harvest, Preparation, and Stratification
- Harvest seeds in the fall (September to October).

- Remove husks from seeds within approximately one month of harvest. It is ideal to remove the husks when they are firm, yet slightly soft; after this point they can become too soft and quite difficult to remove.
-
- First, remove the major portion of the husk using multiple possible methods on a hard surface (driveways, etc.): pounding with a rake, stomping with feet, running over with light or mid-weight vehicles, peeling off with hands, or any other form of abrasion that can safely remove the husks without cracking the shell.

- Immediately follow this by power-washing, if possible, to remove remaining bits of husk from the shells.Remove husks from seeds within approximately one month of harvest. It is ideal to remove the husks when they are firm, yet slightly soft; after this point they can become too soft and quite difficult to remove.

- First, remove the major portion of the husk using multiple possible methods on a hard surface (driveways, etc.): pounding with a rake, stomping with feet, running over with light or mid-weight vehicles, peeling off with hands, or any other form of abrasion that can safely remove the husks without cracking the shell.
-
- Within a few weeks of husk removal, prepare seeds for stratification. Sanitize seeds with a 1:10 bleach to water solution
- Dip and swoosh batches of nuts using a large colander in a bucket of the bleach solution for approximately 15 seconds.

- Rinse under water for approximately 15 seconds.

- Dip and swoosh batches of nuts using a large colander in a bucket of the bleach solution for approximately 15 seconds.
- Pack cleaned seeds in moist, but not wet sand (just enough water so that you cannot squeeze water out of a fistful). Peat, sphagnum moss, or vermiculite can also be used as a stratification medium.
- Make sure that each individual seed is completely surrounded by sand and that a small amount of airflow is able to go through the container – enough that the seeds can respire, but not enough to dry the sand out. This can be facilitated by drilling small holes in the containers or using loose-fitting lids. Inverted plastic cake containers were used here: one to two containers per batch (family). Eight small (3/32”) holes were drilled near the top of each container to allow airflow.

- Make sure that each individual seed is completely surrounded by sand and that a small amount of airflow is able to go through the container – enough that the seeds can respire, but not enough to dry the sand out. This can be facilitated by drilling small holes in the containers or using loose-fitting lids. Inverted plastic cake containers were used here: one to two containers per batch (family). Eight small (3/32”) holes were drilled near the top of each container to allow airflow.
- Store the containers of seeds in a cooler (1-5°C or 34 to 40°F) for stratification.

- Keep the seeds in stratification conditions for 90-120 days.
- Check seeds periodically for mold growth resulting from too much moisture, or to make sure that the sand is not drying out. If the sand is too dry, add water as needed.
Part 2: Seed Germination
Upon completion of stratification period, move the seeds to pre-germination conditions in a warm area, such as a greenhouse, or ideally a growth chamber. If a greenhouse or growth chamber is not available, germination heating mats can also be used and are easily purchased online. While not necessary, pre-germination in warm conditions will encourage much quicker, more uniform germination, and allow for selection of the most viable and healthy seedlings prior to planting into pots or the field.
- Obtain shallow, broad containers or trays (~7” deep) and drill a few small holes in the bottom of the trays to allow for drainage of excess water.

- Fill the trays with moist, but not wet, sand, peat, perlite, vermiculite, and/or soil. A 50:50 sand to perlite mixture was used here.

- Place the seeds in the filled trays. Lay each seed on its side, lengthwise, covering with a small (~1”) layer of substrate, as butternuts have hypogeal (underground) germination. Make sure there is enough room for the radicle to emerge and grow downwards until planting (otherwise, it will grow out horizontally and “tangle” with other roots, making it difficult to extract for planting).

- Cover container to help retain moisture, but still allow a small amount of air flow.
- Place the seed trays into the warm germination area (growth chambers, greenhouse, heating mat, etc.). If possible, aim for environmental conditions as roughly the following:
- Day temperature (8 hours): 86°F
- Night temperature (16 hours): 68°F
- Light is optional during the germination period.

- Check the media frequently to assure a consistent moist, but not wet, moisture level, watering as needed. Make sure to check the seeds at least every 3-4 days to monitor moisture levels, check for germination, and check for any fungal growth. If serious fungal growth occurs, a general pesticide may be applied. Germination was first noted here two weeks after placement of seeds into germination conditions.
- It will generally take approximately 50-80 days for the majority of the seeds to germinate, although a majority of the seeds (64%) germinated by 17 days using the current method. Seeds will begin to germinate by cracking open along the main seam of the seed and very soon afterwards, the radicle will begin to emerge, followed by the hypocotyl and epicotyl.

- Ultimately, a roughly 65% germination rate can generally be expected, although a rate of 86.57% within 45 days was achieved using this method. As each seed germinates and you are able to see the radicle, carefully remove and plant the germinated seedlings, before the fragile radicle extends too much and is more likely to be damaged during transplanting.
This work was funded by the Fred M. van Eck Scholarship through the HITRC. Many thanks to James McKenna and James Warren for their assistance with seed processing and preparation.
Tax Deductions for Casualty Loss of Timber
To say parts of the country have experienced some extreme weather and fire events this year would be a profound understatement. These events can be particularly hard on forest and tree plantation owners as wind, water, fire and ice can all damage or destroy trees. The long-term nature of growing and managing trees for timber makes these events particularly devastating. If you have experienced casualty losses of timber trees – trees destroyed by these events, you may be eligible for an income tax deduction.
Determining how much you may be able to deduct from your taxes will require collecting some information on your timber investment. You will need to determine the adjusted basis of your trees at the time of the loss. Basis represents your costs to acquire the asset if you purchased it, or the fair market value at the time of transfer if inherited. For gifted properties, the new owner’s basis is generally the adjusted basis of the donor. Adjusted basis refers to any additions or subtractions to basis during your ownership, like purchase of additional property or sale of timber.
Deductible casualty loss for timber held for investment of business purposes is the adjusted basis of the timber, or the difference between the fair market value immediately before and after the casualty event, whichever is less. This means you will also need an estimate of fair market value for the destroyed trees before and after the event. Professional foresters can assist landowners in establishing basis and fair market values. If you decide to file for a casualty loss, you must normally claim the deduction on the tax return of the year the casualty occurred.
This article represents a short summary of considerations for claiming a casualty loss, and should not be considered tax advice. For more detailed information on the tax treatment of timber, consult the National Timber Tax website at www.timbertax.org and your tax advisors.
If you have sold timber or planted trees for timber production in 2018, there are additional steps you may take to potentially reduce you tax liability. Check out the publication Tax Tips for Forest Landowners for the 2018 Tax Year at https://timbertax.org/publications/fs/taxtips/TaxTips2018%20(006).pdf
An Update on Chestnut Blight Research at the HTIRC
by Shaneka Lawson1, Aziz Ebrahimi2, and Jim McKenna1
1USDA Forest Service, Northern Research Station, West Lafayette, IN.2 Department of Forestry & Natural Resources, HTIRC, Purdue University, West Lafayette, IN.
The American chestnut tree (Castanea dentata) was once common across the eastern United States and was a dominant forest tree. Their fruit was referred to and popularized by Nat King Cole in the hit song “Chestnuts Roasting On an Open Fire” written in 1945 by Bob Wells and Mel Tormé. In addition to the copious amount of fruit provided by the tree, it was also valued for its decay-resistant wood. Introduction of the invasive fungal pathogen Cryphonectria parasitica from Asia brought about the widespread infection termed “chestnut blight” which devastated North American chestnut populations. The C. parasitica fungus enters the tree through cracks in the bark and grows unchecked throughout the cambium causing necrotic depressions or “cankers” along the way. Eventually, the cankers girdle and kill the tree as they span its circumference. Chinese chestnut (C. mollissima) are resistant to blight as they co-evolved with the pathogen while our American chestnuts are highly susceptible. Fungi within infected trees produce spores that are rapidly spread by wind, rain, and insects to other trees.
Chestnut blight research continues as we work to breed resistant stock and in addition, began a new investigation into biological control of the disease. We found a pure American orchard in northern Indiana where the disease stopped 5 years ago and the trees resumed healthy normal growth (Becky’s Orchard). Just 20 miles north, a second planting of the same seedlings was equally infected 5 years ago and every tree died back to the ground 3-years ago (Cliff’s Orchard). Hypovirulence, where the blight fungus becomes infected itself with a virus, has been demonstrated to reduce the virulence of the pathogen. Rather than girdle the entire stem, hypovirulent cankers grow much smaller and are not lethal, and could explain the recovery of Becky’s Orchard.
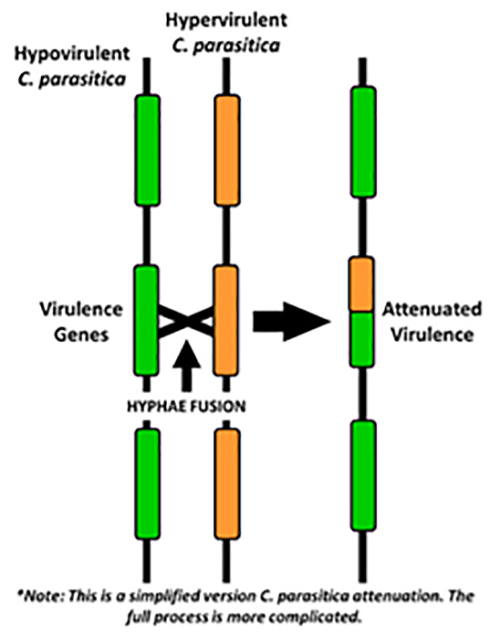
At present, we have confirmed many wild-type pathogenic isolates from Cliff’s Orchard but none from Becky’s. The only fungi recovered at Becky’s were Pestalotia spp. and other saprophytes identified by the Purdue Plant Diagnostic Lab (PPDL). Last December, Mark Ball at the University of West Virginia cultured our samples that were collected in the fall but found none to be hypovirulent.

We conducted in vitro competition experiments on several of Becky’s isolates against wild-type pathogenic blight but our results were inconclusive. Our September collection at Becky’s was higher into the canopy from younger branch crotches and healed blight margins with increased field sterilization to limit saprophytes. This more intensive sampling worked well but alas we still failed to find a hypovirulent isolate (CpHV). Our primary goal is to utilize a biocontrol method to preserve American chestnut for resistance breeding. Thanks to several funding sources and cooperators we have the resources to continue this work to hopefully find CpHV for Indiana landowners working toward our ultimate goal: to conserve and restore American chestnut.
ACKKNOWLEDGEMENTS We thank the Indiana Nature Conservancy (IN-TNC) for supporting HTIRC’s program for American chestnut tree improvement. We thank Tom Creswell at the PPDL and Mark Ball at the University of West Virginia for identifying isolates; Jason Cook, Arbor America Inc.; the Indiana Chapter of the American Chestnut Foundation (IN-TACF) and USDA Forest Service, Northern Research Station and the Forestry & Natural Resources Department at Purdue University for their ongoing support.
Thousand Cankers Disease Best Management Practices Recommendations
The following best management practice recommendations are derived from an article in The Woodland Steward Fall Issue 2018 (Volume 27, Number 2). Although the article targets Indiana, these best management practices have broad application across the central hardwoods region for landowners planting and managing black walnut. You may view the full article here
Thousand Cankers Disease: An Indiana Perspective
by Holly Wantuch and Matthew Ginzel
Thousand Cankers Disease (TCD) is a disease of black walnut and some other walnut species carried by the walnut twig beetle (WTB), Pityophthorus juglandis, boring into walnut bark, and caused by the fungus Geosmithia morbida, which forms cankers in the bark.
Trees under physiological stress are more susceptible to pests and pathogens and are more attractive to wood-boring beetles. As such, effort should be aimed at keeping trees healthy and monitoring closely for TCD and WTB. With these circumstances in mind, we suggest the following best management strategies for walnut growers in Indiana:
- Continue TCD monitoring efforts, especially at the sites most likely for introduction (e.g., mills, yard waste sites, land-clearing businesses, etc.).
- Maintain tree health and vigor.
For plantations:
- Establish trees only on well-suited sites.
- Manage weeds, especially in the early years.
- Maintain appropriate thinning practices and schedule, girdling non-crop trees in August or September.
- Monitor girdled trees for WTB.
- If WTB is found in your area, girdled trees should be removed from the plantation and destroyed. (Do this only on days with temperatures below 50°F.)
- Maintain appropriate pruning practices.
- We do NOT suggest the pre-salvage harvest of walnut in Indiana at this time.
For urban areas:
- Plant black walnut only on suitable sites.
- Beware of changing soil grade or adding fill around walnut.
- During drought periods, provide trees supplemental water.
Natural stands:
- Use Timber Stand Improvement (TSI) techniques to maintain walnut health.
- Monitor crown condition of crop trees annually. If crown deterioration (increased dieback) is observed, contact state or consulting forester for assistance.
Currently TCD does not pose an imminent threat to black walnut production in Indiana. Geosmithia morbida is a weak fungal pathogen and mass attack by the beetle is required to introduce the amount of fungus necessary to kill a susceptible tree. Also, studies of TCD outbreaks in the eastern US suggest that the disease does not impact the trees in their native range as drastically as it has been seen to in the West. For instance, black walnut trees experiencing TCD-related decline in Tennessee and Virginia actually recovered from the disease – an outcome far different from that seen the western US. Likewise, researchers at Purdue have also monitored the health of trees at TCD outbreaks in Ohio since 2015 and have observed no disease progression among those trees.
Dr. Holly Wantuch is a post-doctoral researcher in the Entomology Department at Purdue University. Dr. Matthew Ginzel is an associate professor of forest entomology. Dr. Ginzel has a joint appointment with the Departments of Entomology and Forestry & Natural Resources at Purdue University. Both authors are part of a team of researchers with Hardwood Tree Improvement and Regeneration Center, https://htirc.org//. The mission of the HTIRC is to advance the science and application of tree improvement, management, and protection of hardwood forests.
Applications of Pest Monitoring Data in Intensively Managed Walnut Plantations
By Geoff Williams1 and Matthew Ginzel2
1PhD Student and Fred M. Van Eck Scholar
2Associate Professor of Forest Entomology and Co-Director of HTIRC
Ambrosia beetles are small, cryptic, and opportunistic pests that can cause significant damage in nurseries, plantations, and natural forests. They are cryptic because they spend the majority of their lifecycle developing inside of the host tree, making them difficult to manage. Ambrosia beetles are opportunistic because they typically exploit trees that are already in a state of physiological stress.
Most ambrosia beetles have a wide host range. Adult female beetles locate a susceptible host by honing in on smells released by stressed trees, such as ethanol. Once there, they tunnel into the sapwood and heartwood and cultivate a fungal garden on which to rear their offspring. Ambrosia beetles get their name from the fact that they feed entirely on these ‘ambrosia’ fungi, which in turn feed on the tree itself. The larvae complete development and typically overwinter in the host tree. Adults emerge in the spring to find a new host. Many bark and ambrosia beetles often produce a second generation of adults within one growing season, in the late summer to fall.
At high populations levels, ambrosia beetles can significantly reduce the quality and quantity of timber produced in plantations. Two common invasive ambrosia beetles, Xyleborinus saxesenii, a native of Europe, and Xylosandrus crassiusculus, a native of tropical and subtropical Asia (Figs 2 & 3) are particularly problematic in plantations of fine hardwood trees in the Central Hardwood Forest Region. Nevertheless, Integrated Pest Management (IPM) tactics, including early detection and monitoring of pest population levels, can trigger timely management actions to keep population levels low.
Due to their cryptic nature, effective management of ambrosia beetles relies heavily on a strong understanding of their ecology and biology, particularly their dispersal behavior. Armed with this knowledge, managers can implement treatment and management efforts when and where they will be most effective at reducing pest populations. In an attempt to develop novel, biologically-informed IPM approaches for ambrosia beetles in hardwood plantations, we explored the extent to which climatic and spatial variables could be used to predict when and where ambrosia beetle populations would be highest in intensively cultivated walnut plantations. In particular, we used long term monitoring data to determine the extent to which ambrosia beetle populations were influenced by A) proximity to woodlots and forests that could provide additional resources outside the plantation, or B) climatic conditions during the previous growing season and winter.
HTIRC stakeholder Arbor America, Inc. owns and manages 620 acres of intensively managed plantations of eastern black walnut in Tippecanoe Co. (Fig. 1). As part of an IPM plan for the Arbor America plantings, the Forest Entomology Lab has assisted with weekly monitoring for bark and ambrosia beetles over the course of the last six growing season. Undergraduate technicians and graduate students identified and counted all the beetles from 80 funnel traps (Fig 4) baited with ethanol, on a weekly basis. As a result of this long-term monitoring effort, over 12,000 Xb. saxesenii and nearly 10,000 Xs. crassiusculus were captured in the traps, as well as smaller numbers of over 100 other beetle species.
Our major discovery was that the relative importance of climatic vs. spatial variables as predictors of ambrosia beetle abundance shifted over the course of the growing season. During the spring, temperature and precipitation from the previous growing season and winter were important predictors of ambrosia beetle populations. As may be expected considering the temperate to tropical climates of their native home ranges, highest population levels of Xb. saxesenii and Xs. crassiusculus during the spring flight period followed years with mild, temperate weather. In particular, populations peaked in the spring following a previous growing season with low temperature and high precipitation and a warm and dry winter.
As the season progressed into late summer and fall, proximity to alternate resources outside the plantation provided more predictive power than climatic factors for ambrosia beetle abundance. In particular, proximity to the edges of plantations and proximity to other plantations, woodlots, and continuous forest corridors were strong predictors of ambrosia beetle abundance during the late summer and fall. Edges of hardwood plantations, including the Arbor America walnut plantations, are often protected by windbreaks, or rows of large trees of more hardy species. These windbreaks likely serve as reservoirs for ambrosia beetles, especially considering that windbreak trees are often in a state of constant stress on the edge, for example, from wind exposure or herbicide drift from surrounding agricultural fields (Fig. 5). Because Xb. saxesenii and Xs. crassiusculus can infest hundreds of species of trees, neighboring forests and woodlots could also serve as reservoirs for ambrosia beetles (Fig 6).
Our findings are consistent with the known ecology and biology of ambrosia beetles, but also include new key insights that will be of use to short-rotation hardwood growers who seek to protect fine hardwoods from damage. Specifically, we recommend that growers initiate monitoring early to detect incipient pest populations. Second, following mild weather, conduct sanitation in the late winter and employ preventative chemical treatments in the spring, including active management of windbreaks. Sanitation may consist of pruning dead branches, disposal of debris, and salvage of declining trees to remove host material that could contribute to ambrosia beetle population build-up. Preventative chemical treatment consists of spraying the bark of crop trees with pyrethroids, to protect the trees from ambrosia beetle attacks. Third, windbreaks and neighboring forests and woodlots may serve as reservoirs for ambrosia beetles. Pest management activities in plantations are often cost and/or time limited, so managers should focus efforts to plantation edges and plantations that are connected with outside forest corridors or woodlots to maximize the economic return on their investment. In particular, the active management of windbreak trees could be an effective tactic to prevent ambrosia beetle population build-up.
In the future, we hope to see more of these types of monitoring data collected in plantations of walnut and other fine hardwoods. Additional data would provide opportunities to further refine our models as they relate to climatic and spatial variation. Furthermore, the approach used in this study could be applied to analyze the effects of management history and intensity on pest populations, to evaluate the effectiveness of our management recommendations. The Forest Entomology Lab thanks Guillermo Pardillo, Arbor America, Inc., and a crew of graduate and undergraduate researchers for their assistance in the collection of the data for this study.
Figure 1. Arbor America, Inc.

Figure 2. Xyleborinus saxeseni

Figure 3. Xylosandrus crassiusculus

Figure 4. Lindgren multi-funnel trap baited with an ethanol lure used to intercept adult ambrosia beetles during their flights to a new host tree. Beetles are attracted to the ethanol, fly into the sides of the funnels, and fall into the collection cup at the bottom. Cups were emptied weekly, and all beetles identified and counted in the laboratory.

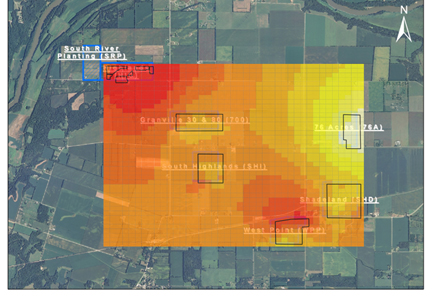
Figure 5. Representative spatial distribution of Xylosandrus crassiusculus in one of the Arbor America, Inc. South Highland walnut plantation. Blue triangles indicate locations of monitoring traps. Red indicates highest number of beetles, and yellow indicates smallest number of beetles. Note the higher density of beetles near the plantation edges.
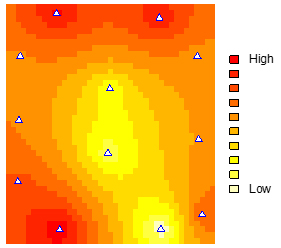
Figure 6. Heatmap of density of spring Xylosandrus crassiusculus populations overlaid on an aerial photograph of all six Arbor America, Inc. walnut plantations. Plantations are denoted with black polygons overlaid on the heatmap. Red indicates highest number of beetles, and yellow indicates smallest number of beetles. Note that populations are highest near the continuous forest corridor which follows the Tippecanoe River (NW corner) and other woodlots (SE corner).
HTIRC Partner Highlighting Their Work on Facebook
The research and outreach work of HTIRC is enhanced by partnerships with businesses, agencies, and individuals across the nation. In 2008 and 2009 the Forgey family began a journey to plant large areas of their Rush County, Indiana farmland to hardwood trees. Mike Forgey approached the HTIRC to see if we would be interested in using some of his family farmland for our tree breeding plantings. Many of the soil types on the Forgey farm were well-suited to a variety of hardwoods, so it was a mutually beneficial arrangement. Parts of the farm were planted to HTIRC hardwood tests and additional acreage was entered into the USDA Conservation Reserve Program and planted to mixed hardwoods. Since those early days, Mike’s son Aaron has graduated from Purdue University with a degree in forest management and established a business to license and sell trees originating from the HTIRC breeding programs. Several additional cooperative projects and events have occurred, including a 2014 forestry field day using the Forgey Tree Farm as a demonstration tour area for local landowners and school children.
The Forgey Tree Farm has a Facebook page highlighting their activities. If you would like to see photos and learn more about Forgey Tree Farm, visit their Facebook site at:
https://www.facebook.com/groups/143995779551315/
 Mike Forgey enjoying a moment at the 2014 field tour at the Forgey Tree Farm
Mike Forgey enjoying a moment at the 2014 field tour at the Forgey Tree Farm
HTIRC Tree Improvement 2018 Seed Harvest and Distribution of Seed
by Jim McKenna, USDA Forest Service, Operational Tree Breeder
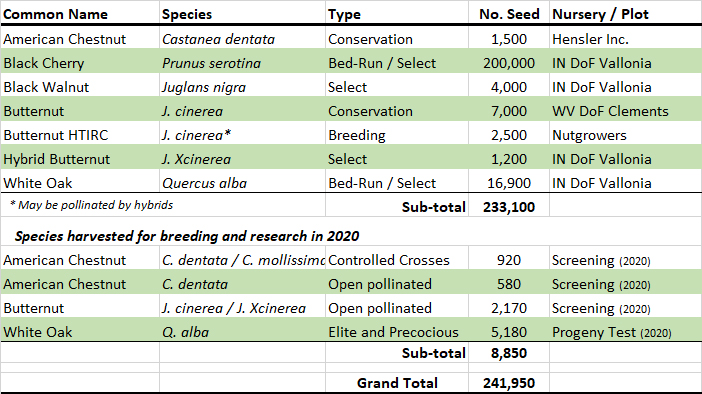
The 2018 HTIRC harvest totals are presented in the table above along with distribution of extra harvested seed to our cooperators. Our chestnut crosses consisted of new F1 hybrids of American by Chinese. We crossed 19 trees and produced 15 new F1 lines. The Americans and Chinese are trees unique to Indiana and Purdue. Hensler Nursery Inc., in Indiana has begun growing pure American chestnuts from our 2010 grafted Duke pure American orchard in partnership with the IN-The American Chestnut Foundation (TACF) and the national TACF.
We harvested select and bed run cherry seed this season from two grafted cherry blocks, one 20 and the other 10-years old and collected just over 200,000 seed to make up for a shortfall in the crop in southern Indiana experienced by our cooperators with the Vallonia Nursery, Indiana Division of Forestry. The walnut crop was down this year and we were only able to collect some advanced selections from a few orchards at Martell that we provided to Vallonia as well.
Our two pure butternut seed orchards both produced about 4,000 seed each this year and we arranged for the Clements nursery (West Virginia Division of Forestry) to grow and distribute the seedlings to landowners in the target areas of the Wayne and Hoosier National Forests in southern Ohio and Indiana, and elsewhere starting in 2020. Extra hybrid butternut from our new resistant orchard at the Pinney Purdue Agricultural Center, Martell germplasm block, and butternut canker screening blocks was provided to the Vallonia Nursery for their hybrid butternut production. Extra pure butternut seed, some likely pollinated from hybrids, will be provided to our neighboring nutgrowers associations.
Our two grafted white oak orchards at Purdue produced very well, both established 10 years ago in 2008. We sowed 300 acorns of 19 families that produced acorns. Our Elite orchard comprised of timber selections produced over 14,000 acorns and our Precocious white oak orchard produced 12,000. The Elite orchard has more parents and is twice the size of the Precocious orchard. On a per-acre basis, our Precocious orchard produced 8,020 acorns / acre while the Elite orchard produced 4,600 / acre. Both orchards are only half grafted. Of the seedling rootstocks, only some of swamp white oak rootstocks have produced acorns so far.
New HTIRC Research Grants to Advance Strategic Plan Goals
HTIRC leadership accepted proposals for research projects to advance the goals of the new HTIRC strategic plan. The HTIRC Executive Committee reviewed these proposals and decided to approve seven of the projects for funding. The titles and principal investigators of the selected proposals are:
Title: Improving establishment practices of pure and mixed hardwood plantations by refining soil suitability indices for black walnut
- Shaneka Lawson (PI)
- James Warren (Co-PI)
Title: Characterizing abiotic and biotic tree stress using hyperspectral information
- John J Couture (PI
- Douglass Jacobs (Co-PI)
Title: Tree Inventory with Aerial Remote Sensing
- Songlin Fei (PI)
- Guofan Shao (Co-PI)
External collaborators
- Joseph P Hupy, Associate Professor, School of Transportation and Aviation Technology, Purdue University, jhupy@purdue.edu
- Joey Gallion, Forest Inventory Program Manager, Indiana DNR
Title: Using terrestrial laser scanning to assess tree health and quality
- Brady S. Hardiman (PI)
- Songlin Fei (co-PI)
Title: Productivity-diversity relationships in hardwood plantations
- Douglass F. Jacobs (PI)
- John Couture (Co-PI)
- Lenny Farlee (Co-PI)
- Brady Hardiman (Co-PI)
- Gordon McNickle (Co-PI)
Title: Understanding and manipulating plant-soil feedbacks to manage the invasive shrub Lonicera maackii
- Michael Jenkins (PI)
- Pierre-Luc Chagnon (co-PI), Université de Montréal, Institut de Recherche en Biologie Végétale, 4101 Sherbrooke Est, Montréal (QC) H1X 2B2 Canada, pierre-luc.chagnon@umontreaca
- Lenny Farlee (co-PI)
Title: Natural and artificial regeneration growth response of white oak across light and understory competition gradients
- Michael R. Saunders (PI)
- Mark Coggeshall (Co-PI)
External collaborator:
Trent Osmon, Installation
Conference Announcements
35th Southern Forest Tree Improvement Conference (SFTIC), June 3-6, 2019,Lexington, Kentucky
The 2019 SFTIC will be hosted by the University of Kentucky, Department of Forestry and Natural Resources, the USDA Forest Service, Southern Research Station, the Kentucky Division of Forestry and the Forest Health Research and Education Center (www.foresthealthcenter.org ). The meeting will run from Monday afternoon through Wednesday evening, with an optional white oak genetics symposium and field tour on Thursday. More details to come and at www.sftic.org . We look forward to seeing you all next June in Kentucky.
Honors and Graduations
Dr. Douglass Jacobs received the Corinne Alexander Sprit of the Land Grant Mission Award in November 2018. The annual award, established in 2008, recognizes faculty members in the Purdue University colleges of Agriculture, Veterinary Medicine and Health and Human Sciences working across all three land-grant mission areas. Two years ago, the award was named in honor of Corinne Alexander, a professor in the Department of Agricultural Economics, who died unexpectedly in 2016. Alexander’s work demonstrated a fierce commitment to the values of land-grant institutions.
“Douglass Jacobs’ outstanding work in integrated research, extension, and education truly represents the spirit of the land-grant mission institutions,” says Karen Plaut, the Glenn W. Sample Dean of the Purdue College of Agriculture. “His work is making a difference in the hardwood tree industry in Indiana as well as across the country.” The full news release is available here
Dr. Songlin Fei, Forestry and Natural Resources: 2018 University Faculty Scholar – Purdue University. The University Faculty Scholars Program “recognizes outstanding faculty members at the West Lafayette campus who are on an accelerated path for academic distinction. Eligible faculty must hold the rank of tenure associate or full professor and have been in that rank for no more than five years. Faculty Scholars are appointed for a nonrenewable five year term and receive an annual discretionary allocation.” The full announcement is available here
Dr. Shaneka Lawson, US Forest Service and Purdue Forestry and Natural Resources: Unsung Diversity Hero Award – College of Agriculture, Purdue University. The “Unsung” Diversity Hero award recognizes faculty, staff, and students who have positively influenced a program, department, or organization from behind the scenes with a positive attitude, a willingness to help in whatever capacity necessary, and a commitment to excellence.
Summer and Fall 2018 Graduates:
Congratulations to our recent graduates: Emily Thyroff, Weston Schempf, Meghan Mulroy, and Mariam Valladares. We wish them the best!
2018 Research Publications
HTIRC-related research papers published in 2018 are listed and linked below. To see a listing of research from previous years, please visit the HTIRC website “Resources” tab: https://htirc.org//research/research-publications/
- Pooled whole-genome sequencing of interspecific chestnut (Castanea) hybrids reveals loci associated with differences in caching behavior of fox squirrels (Sciurus niger L.) (PDF 648 KB) 2018. LaBonte, N.R. and Woeste, K.E. Ecology and Evolution DOI: 10.1002/ecew.4336.
- Short-term vegetation Responses to Invasive Shrub Control Techniques for Amur Honeysuckle (Lonicera maackii[Rupr.] Herder) (PDF 34 KB) 2018. Frank, G.S., Saunders, M.R., and Jenkins, M.A. Forests 9(10):DOI: 103390/19100607.
- Natural regeneration of Pinus pinaster facilitates Quercus ilex survival and growth under severe deer browsing pressure (PDF 11 KB) 2018. Malton, A., Mariotti, B., Tani, A., Martini, S., Jacobs, D.F., and Tognetti, R. Forest Ecology and Management 432(01):356-364.
- Soil chemistry and microbial community functional responses to invasive shrub removal in mixed hardwood forests. (PDF 10 KB) 2018. Frank, G.S., Nakatsu, C,H., and Jenkins, M.A. Applied Soil Ecology 131:75-88.
- Resources for studies of iron walnut (Juglans sigillata) gene expression, genetic diversity, and evolution (PDF 1 MB) 2018. Feng, X., Yuan, X., Sun, Y., Hu, Y., Zulgigar, S., Quyang, X., Dang, M., Zhou, H., Woeste, K,, Zhoa, P. Tree Genetics & Genomes 14:51 DOI: 10.1007/s11295-018-1263-z
- Linking Landsat to terrestrial LiDAR: Vegetation metrics of forest greenness are correlated with canopy structural complexity (PDF 34 KB) 2018. LaRue, Atkins, J.W., Dahlin, K., Fahey, R., Fei, S., Gough, C., and Hardiman, B.S. International Journal of Applied Earth Observation and Geoinformation 73:420-427.
- Allocation of forest biomass across broad precipitation gradients in China’s forests (PDF 10 KB) 2018, Lie, Z., Xue, L., and Jacobs, D.F. Scientific Reports DOI: 10.1038/s41598-018-28899-5.
- Endogenous translocation patterns of current photosynthate in post-transplant Quercus ruba seedlings (PDF 34 KB) 2018. Sloan, J.L., Jackson, G.S., and Jacobs, D.F. Canadian Journal of Forest Research DOI: dx.doi.org/10.1139cjfr-2018-0087.
- Genetic diversity of superior Persian walnut genotypes in Azadshahr, Iran (PDF 532 KB) 2018. Shamlu, F., Rezaei, M., Lawson, S., Ebrahimi, A., Biabani, A., and Khan-Ahmadi, A. Physiology and Molecular Biology of Plants DOI https://doi.org/10.1007/s12298-018-0573-9.
- Using a tree seedling mortality budget as an indicator of landscape-scale forest regeneration security (PDF 193 KB) 2018. Vickers, L,A., McWilliams, W.H. ,Knapp, B.O., D’Amato, A.W., Saunders, M.R., Shifley, S.R., Kabrick, J.M., Dey, D.C., Larsen, D.R., and Westfall, J.A. Ecological Indicators DOI https://doi.org/10.1016/j.ecolind.2018.06.028.
- Overstory species response to clearcut harvest across environmental gradients in hardwood forests (PDF 233 KB) 2018. Swaim, J.T., Dey, D.C., Saunders, M.R., Weigel, D.R., Thornton, C.D., Kabrick, J.M., and Jenkins, M.A. Forest Ecology and Management 428:66-80.
- Genetic engineering of trees: progress and new horizons (PDF 1 MB) 2018. Chang, S., Mahon, E.L., MacKay, H.A., Rottmann, W.H., Strauss, S.H., Pijut, P.M., Powell, W.A., Coffey, V., Lu, H., Mansfield, S.D., and Jones, T.J. In Vitro Cellular and Developmental Biology-Plant DOI: 10.1007/s11627-018-9914-1.
- Optimization of Agrobacterium-mediated genetic transformation of Fraxinus nigra and development of black ash for possible emerald ash borer resistance (PDF 1,019 KB) 2018. Lee, J.H. and Pijut, P.M. Plant Cell, Tissue and Organ Culture 134(2):217-229.
- Promoting and maintaining diversity in contemporary hardwood forests: Confronting contemporary drivers of change and the loss of ecological memory (PDF 227 KB) 2018. Webster, C.R., Dickinson, Y.L., Burton, J.I., Frelich, L.E., Jenkins, M.A., Kern, C.C., Raymond, P, Saunders, M.R., Walters, M.B., and Willis, J.L. Forest Ecology and Management 421:98-108.
- Enhanced forest interior estimations utilizing lidar-assisted 3D forest cover map (PDF 10 KB) 2018. Shao, G., Iannone III, B.V., and Fei, S. Ecological Indicators 93:1236-1243.
- Spatial Variation in Canopy Structure across Forest Landscapes (PDF 34 KB) 2018. Hardiman, B.S., LaRue, E.A., Atkins, J.W., Fahey, R.T., Wagner, F.W., and Gough, C.M. Preprints 2018060351 (doi: 10.20944/preprints201806.0351.v1).
- Signatures of Selection in the Genomes of Chinese Chestnut (Castanea mollissima Blume): The Roots of Nut Tree Domestication (PDF 257 KB) 2018. LaBonte, N.R., Zhao, P., and Woeste, K. Frontiers in Plant Science 9:1-13.
- Data mining for discovery of endophytic and epiphytic fungal diversity in short-read genomic data from deciduous trees (PDF 257 KB) 2018. LaBonte, N.R., Jacobs, J., Ebrahimi, A., Lawson, S., and Woeste, K. Fungal Ecology 35:1-9.
- Dominant forest tree mycorrhizal type mediates understory plant invasions (PDF 10 KB) 2018. Jo, I., Potter, K.M., Domke, G.M., and Fei, S. Ecology Letters 21(2):217-224.
- Metrics for macroscale invasion and dispersal patterns (PDF 10 KB) 2018. Clark, T.L., Iannone, III, B.V., and Fei, S. Journal of Plant Ecology 11(1):64-72.
- Behavioral Responses of Pityophthorus juglandis (Coleoptera: Curculionidae: Scolytinae) to Volatiles of Black Walnut and Geosmithia morbida (Ascomycota: Hypocreales: Bionectriaceae), the Causal Agent of Thousand Cankers Disease (PDF 59 KB) 2018. Blood, B.L., Klingeman, W.E., Paschen, M.A., Hadžiabdić, Đ, Couture, J.J., and Ginzel, M.D. Environmental Entomology 47(2):412-421.
- Reflectance spectroscopy: a novel approach to better understand and monitor the impact of air pollution on Mediterranean plants (PDF 11 KB) 2018. Cotrozzi, L., Townsend, P.A., Pellegrini, E., Nali, C., and Couture, J.J. Environmental Science and Pollution Research 25(9):8249-8267.
- Shifting conceptions of complexity in forest management and silviculture (PDF 34 KB) 2018. Faheya, R.T., ,Alvesherea, B.C., Burtonb, J.I., D’Amatoc, A.W., Dickinsond, Y.L., Keetonc, W.S., Kerne, C.C., Larsonf, A.J., Palikg, B.J., Puettmannh, K.J., Suanders, M.R., Websterd, C.R., Atkins, J.W., Gough, C.M., and Hardiman, B.S. Forest Ecology and Management 412: 59-71.
- Maintaining Indiana’s Urban Green Spaces: A Report from the Indiana Climate Change Impacts Assessment (PDF 34 KB) 2018. Reynolds, H., Brandt, L., Widhalm, M., Fei, S., Fischer, B., Hardiman, B., Moxley, D., Sandweiss, E., Speer, J., and Dukes, J. Urban Green Spaces Reports. Paper 1.
- Improving Lidar-based aboveground biomass estimation of temperate hardwood forests with varying site productivity (PDF 43 KB) 2018. Shao, G., Shao, G., Gallion, J., Saunders, M.R., Frankenbert, J.R., and Fei, S. Remote Sensing of Environment 204:872-882.
- Blends of Pheromones, with and without Host Plant Volatiles, Can Attract Multiple Species of Cerambycid Beetles Simultaneously. (PDF 34 KB) 2018. Hanks, L.M., Mongold-Diers, J.A., Atkinson, T.H., Fierke, M.K, Ginzel, J.D., Graham, E.E., Poland, T.M., Richards, A.B., and Millar, J.G. Journal of Economic Entomology 3(2):716-724.
- Localized gene expression changes during adventitious root formation in black walnut (Juglans nigra L.) (PDF 1 MB) 2018. Stevens, M.E., Woeste, K.E. and Pijut, P.M. Tree Physiology 38:877-894.
- Rapid in vitro Shoot Multiplication of the Recalcitrant Species Juglans nigra L. (PDF 503 KB) 2018. Stevens, M.E. and Pijut, P.M. In Vitro Cellular & Developmental Biology-Plant 54:309-317.
- Population genetics, phylogenomics and hybrid speciation of Juglans in China determined from whole chloroplast genomes, transcriptomes, and genotyping-by-sequencing (GBS) (PDF 1MB) 2018. Zhao, P., Zhou, H.J., Potter, D., Hu, Y.H., Feng, X.J., Dang, M., Feng, L., Zulfiqar, S., Liu, W.Z., Zhao, G.F., and Woeste, K. Molecular Phylogenetics and Evolution 126:250-265.
