HTIRC E-NEWSLETTER – May 2013
Volume 6 Issue 1
Articles in this issue:
- Butternut Overview
- Disease Progression in Slocum’s Woods
- Butternut Canker Resistance and Novel Screening Procedures
- Evaluating the Ecology of Butternut and Naturally Occurring Hybrids with Japanese Walnut
- Millions of North American and European Ash trees Face Certain Death
- Epicormic Branching in Oaks: Three Year Results
- Indiana’s Maples May be Next on the Menu
- From Zamorano, Honduras to West Lafayette, Indiana – HTIRC Comtemplates a Dual-Language Intitiative!
- Thousand Cankers Disease Webinars Provide New Information for 2013
Welcome to this issue of the Hardwood Tree Improvement and Regeneration Center E-newsletter. The HTIRC is committed to enhancing the productivity and quality of Central Hardwood Region trees and forests for the economic and environmental benefits they provide. Scientists at the HTIRC are using conventional tree improvement breeding as well as molecular and genetic technologies to improve the wood quality, growth characteristics, and insect and disease resistance of trees like black walnut, black cherry, red and white oaks, butternut and American chestnut.
Research in tissue culture, tree nursery practices, tree plantation establishment and management, and Central Hardwoods silvicultural systems is aimed at increasing the regeneration success rate for high quality hardwood trees and forests. Some interesting and unusual research areas include examining the potential for propagating trees with “figured” wood: birds-eye maple or curly walnut; and breeding trees that will be an economical source of bio-fuels.
Twice per year we will attempt to provide interesting and useful information on Central Hardwood trees and forests, as well as sources for additional information and assistance.
Please pass this newsletter along to others who may enjoy or benefit from the information provided. If you would like a closer look at the HTIRC, please visit our web site at: http://www.htirc.org
Butternut Overview
For more than a decade, HTIRC has been working to restore butternut (Juglans cinerea L.) as part of its mission to improve and conserve fine hardwood species. This relatively unknown and underutilized hardwood species was once ranked second for veneer production, behind black walnut (J. nigra L.), in Indiana. Unfortunately butternut canker disease, caused by the fungus Ophiognomonia clavigignenti-juglandacearum (Oc-j) and believed to be an exotic pathogen, has severely affected butternut. Butternut canker was first identified by scientists in 1967, but over the past 40 years or so, butternut populations throughout the range have declined 77%. While some butternuts still remain, recent disease surveys have found greater than 95% infection among surviving individuals in New England. In response to population declines, Canada listed butternut as endangered in 2003. The United States does not list butternut nationally, but 22 of 30 states within butternut’s range list the species as vulnerable or critically imperiled.
Efforts to restore butternut have focused primarily on three areas: identifying resistant trees, determining the genetic viability of the remaining population, and characterizing planting sites where butternut will be most successful. The following sections provide a current and comprehensive review of butternut research conducted at the HTIRC: disease progression in a butternut stand, butternut canker disease pathology and screening of resistant material, and how hybrids identified during initial resistance surveys compare to butternut.
Disease Progression in Slocum’s Woods
By Nick LaBonte, Ph.D. HTIRC Graduate Student
Slocum’s Woods, an unusually dense population of butternuts near Whitewater, WI, has been the subject of butternut conservation and butternut canker research since its discovery in the early 1990s. At its peak, sometime before 2001, over 600 individuals populated the 38-acre woodlot As of May 2012, only 113 living trees remain. Most of the mortality can be attributed to butternut canker disease, but some trees in the population have remained remarkably healthy. In addition, some butternuts in the population have unusual darkly-colored bark, which is believed to be associated with canker resistance. Do the surviving trees have some genetic basis for disease resistance? If they do, they could be bred with other resistant butternuts to produce canker-resistant butternut planting stock.
We used 21 DNA-based markers (microsatellites) to determine which butternuts in the population were relatives and which were unrelated. This large number of markers gave us great power to distinguish related and unrelated trees. If there is a strong genetic basis for canker disease resistance, we would expect the closely related trees to have similar disease symptoms in the field. Similarly, if the dark bark trait, previously suggested as an indicator of disease resistance, has a genetic basis, we would expect relatives to have more similar bark color than unrelated trees.
We assessed disease severity in the field with six measures: number of cankers below 1.5 m, number of cankers at the soil line, percent of major limbs dead, canker virulence (based on the level of canker containment and healing), crown vigor, and the presence of epicormic branches. These measures were combined into an index where the healthiest trees had the lowest score, and badly cankered trees had the highest scores. This index of disease resistance was strongly correlated with bark color, with darker-barked trees tending to have lower disease index scores. However, when relatedness was compared with disease severity and bark color, related trees were not significantly more likely than unrelated trees to have similar disease scores or bark color. There were significant, but weak, correlations between relatedness and the number of cankers and percent of crown limbs dead. These results indicate there might be some genetic basis for canker resistance in butternut, but it may be too weak to be of value for tree breeders.
We used additional data to determine other factors that might have contributed to butternut decline in Slocum’s Woods. In many plant disease systems, subtle environmental factors affect disease impacts and mortality. We hypothesized that environmental conditions would have a significant impact on tree mortality in Slocum’s Woods over a ten year-period. We used a butternut census of the site from 2001 (with GPS coordinates) to identify trees that had survived to the present. Combining tree location data with a soil map allowed us to split the Slocum’s Woods population into two groups. One occupied a limestone knoll “micro-site” with about two feet of topsoil and the other, a flat ancient lakebed where soils are deep, silty loams over sand and gravel. We analyzed the influence of bark color and soil type on a tree’s probability of survival over the ten-year period. Dark-barked trees, and those growing on the thinner, rocky soils, were about twice as likely to live as light-barked trees and those on deep silt soils. This evidence correlates well with research showing that butternut seedlings are flood intolerant. Historical accounts of butternut prevalence and data from recent HTIRC research indicate that planting efforts for butternut restoration might have a greater chance of success on upland sites.
Butternut Canker Resistance and Novel Screening Procedures
By James J. Jacobs, Ph.D. HTIRC Graduate Student
In the Spring 2012 newsletter we described the development of a foliar inoculation test to help identify resistant and susceptible butternut trees. Since then, preliminary data has confirmed colonization of butternut and hybrid leaves in the laboratory is due to Oc-j (the fungus that causes butternut canker) and the inoculation protocol has been refined. Large necrotic lesions are readily formed in moisture chambers when detached leaflets are wounded, and even within tissue undamaged by wounding. Images collected using scanning electron microscopy (SEM) indicate the fungus is able to grow into leaf tissue through stomata, without wounding. It also appears that the fungal germination patterns on leaflet and stem tissue differ. This may be attributable to different chemicals produced by the tree in stem tissue versus foliar tissue.
Preliminary results indicate that the expansion of leaf lesions varies between seed families (seed collected from different mother trees). This differential severity followed patterns observed in field stem inoculations of these families and may indicate susceptibility to the fungus in foliar tissues is genetically inherited. Sadly, few stem cankers resulted from inoculations in 2011 and we were unable to perform the foliar-stem correlative analysis initially proposed. Therefore, in 2012, we conducted another test using five seed families of butternut, hybrid trees, and Japanese walnut. To increase the percentage of canker “take” on stem tissue we inoculated these trees with sterilized grain (oat and rye) colonized by Oc-j, instead of the traditional inoculation method using agar plugs containing Oc-j. We hypothesized greater inoculation success with grain compared to agar because grain provides a readily available food source for the fungus and increases the moisture available for fungal colonization of stem tissue, thereby increasing the chances of successful colonization of the tree stem. If successful, a higher percentage of successful canker inoculations will allow us to distinguish between resistant and susceptible selections by correlating stem canker expansion with foliar disease progression in these seedlings
Sections of cankered stem material from a subset of the trees inoculated in 2012 were collected periodically and are in preparation for histological analysis. Additionally, sections of stem tissue inoculated with spore suspensions have been imaged using SEM. These stem sections have shown that spore germination may be limited on the surface of woody tissue and spore germination patterns indicate that wounding (as previously suggested) may be essential to colonization of stem tissue.
In the Spring 2012 newsletter we also outlined the study of epiphytes (fungi that live outside of the host) and endophytes (fungi that live inside the host). Since then, all of the fungi isolated from butternut, Japanese walnut and a hybrid have been identified. A total of 451 fungal isolates were collected from the three taxa at a site in Rosemount, MN and at Martell Forest near Lafayette, IN. Our results indicate these three taxa harbor similar assemblages of fungi when growing on the same site and in close proximity to one another. This indicates the fungal community occupying each of the trees may be locally derived. Additionally, when the isolated epiphyte or endophyte is placed on an agar plate with an isolate of Oc-j, many of the combinations eventually produce an area between the two species devoid of fungal material. This is not an uncommon occurrence when unrelated fungi attempt to occupy the same space. To more definitively identify potential biocontrol isolates, we used an additional screening study where the endophytic or epiphytic isolate is killed after a predetermined amount of growth, leaving only its metabolic byproducts. Oc-j is then plated onto the agar surface. This method indicates that around 6% of the isolated fungi produced chemicals that are actually able to limit growth of Oc-j. Further research is necessary to ascertain whether this phenomenon is functional in the plant under field conditions or if it is limited to laboratory conditions.
Evaluating the Ecology of Butternut and Naturally Occurring Hybrids with Japanese Walnut
By Philip Crystal, recent M.S. HTIRC Graduate Student
Initial surveys for canker resistant butternuts identified hybrids between butternut and Japanese walnut that might serve as a source of disease resistance for butternut tree improvement. While hybrids can be very similar to butternut in appearance, it is unclear if hybrids can fulfill the ecological role of butternut. As such, I refrain from using the term butternut hybrid, which might imply hybrids are more butternut than Japanese walnut. I also would like to caution readers that while extremely vigorous and productive agricultural hybrids (e.g., corn) are desirable, hybrids between species are not always beneficial. Naturally occurring hybridization has been associated with extinction of species, the development of invasive character, and the formation of new species.
Despite these potential negative impacts, the ecological differences between butternut and hybrids have not been well-studied. Assessment of the potential discrepancies is essential if hybrids are to be used as a substitute for butternut in the event of butternut’s extirpation or as a way to breed for resistance in butternut. Field observations indicate first-generation hybrids grow rapidly and produce abundant seed. As reported in the Spring 2012 newsletter article, when exposed to moisture stress in the greenhouse, random hybrid 1+0 (one-year-old bareroot) seedlings were more susceptible to drought and less susceptible to flood than butternut seedlings. Overall, this indicates hybrids may prefer wetter sites than butternut. As flood and drought stress are influenced by seedling size, the observed stress responses could have been due to size differences between butternut and hybrids, despite their similar age. Either explanation for the difference in response to flood and drought, however, indicates that plant species composition could be altered by general broadcasting of hybrid seedlings.
Butternut is considered the most cold-hardy walnut species and as such, is more ecologically important in the northern part of its range. Japanese walnut, however, reportedly suffers cold damage from early spring leaf flush in the southern parts of butternut’s range. Based on this information, during winter 2011-2012 the cold-hardiness of butternut and hybrids in a field plantation was assessed using fall leaf abscission, winter electrolyte leakage (a measure of how susceptible dormant trees are to cold damage), and spring leaf flush. Few differences were detected between families of butternut or hybrids. When grouped into butternut versus hybrids, however, hybrids lost leaves later in the season and leaves were still attached at the first frost. Winter electrolyte leakage revealed hybrids were more susceptible to cold damage during dormancy. No differences were detected in spring leaf flush, the most important criteria for matching plants to the environment. This was probably because remarkably warm days (+80°F) in March helped release all trees at essentially the same time. Overall, hybrids’ later leaf drop may affect nitrogen cycling in natural stands, and reduced tolerance to cold temperatures during dormancy may limit their survival in the northern portions of butternut’s range, especially during harsh winters.
Hybridization is also associated with an increase in variability, especially when multiple generations of hybrids breed with one another. To assess the overall variability of the hybrid gene pool, butternut, hybrids, Japanese walnut, and black walnut seedlings were grown in the greenhouse in summer 2012 and assessed at two trait suites: vegetative traits (appearance) and ecological traits associated with seedling survival. Analysis revealed that roughly 50% of hybrids resembled Japanese walnut in appearance; the remainder resembled butternut or were intermediate. Roughly 25% of hybrids were ecologically similar to butternut or Japanese walnut at the traits assessed. Interestingly, 35% functioned like black walnut and 15% exhibited novel combinations of the ecological characters. These experiments indicate that in the general pool of hybrids there are individuals whose growth habit and ecological function are unlike those of butternut. Despite discrepancies, some hybrids did look and act like butternut in ecological tests. If hybrids are to be used as an ecological substitute or to breed for resistance, it would be ideal to easily identify canker resistant hybrids that look and act like butternut in the wild.


Mature butternuts in a southern Wisconsin woodlot with mild (left) and severe (right) canker disease symptoms.
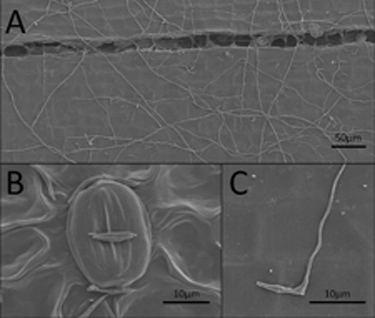
SEM images of butternut tissues inoculated with a concentrated spore suspension
of Oc-j. (A) Growth of fungal hyphae over the surface of stem tissue 200X. (B) Ungerminated spores on leaf perpendicular to closed stomata 1200X. (C) Germinated spore on the surface of the stem 1600X.
Note: 1 µm = 1 micrometer = 1 one-millionth of a meter. A human hair measures between 20 and 120 micrometers

Measuring the appearance and ecological function of butternut, hybrid, Japanese walnut,
and black walnut seedlings. All plants were grown from seed in the greenhouse
for approximately 3 months. For scale, Phil Crystal is roughly six feet tall
Millions of North American and European Ash Trees Face Certain Death
By Shaneka Lawson, Plant Physiologist
Ash trees are hardy, fast-growing hardwoods historically used in reforestation efforts because of their robust growth, high value wood, and ability to survive in diverse environmental conditions. North American ash trees are found primarily in the mid-west and eastern United States with a considerable number of ash trees being found in Canada. Internationally, ash trees have a range that includes the majority of countries in England, Europe, and Asia. Increased threats to ash trees have motivated researchers to find methods for promoting its survival against biotic pest pressure.
In the United States when we hear news regarding ash trees, we tend to zero in on the destruction following in the wake of the Emerald Ash Borer (Agrilus planipennis Fairmaire). Emerald Ash Borer (EAB) is a highly destructive exotic beetle first discovered in Detroit, Michigan and Windsor, Ontario (Canada) in 2002. The adult beetles cause minimal damage when feeding on foliage however the larvae consume both phloem and cambium causing an immediate threat to overall tree survival. There are often numerous EAB larvae within a tree and their continuous feeding in random “S-shaped” patterns will girdle and effectively kill the host tree. These exotic pests likely arrived in the United States in the packing materials aboard ships or planes from Asia. Since 2002, the insects were found in Ohio in 2003, northern Indiana in 2004 and both northern Illinois and western Maryland in 2006. In 2007, the beetle continued its spread across the Midwest and Eastern United States and was discovered in western Pennsylvania and West Virginia. The spread appeared to accelerate as the borer was found in Missouri, Virginia, and Wisconsin in summer of 2008 and in Kentucky, Minnesota and New York in the spring of 2009. Early in 2010 the bug was found in Iowa before being found in Tennessee in late summer. By 2012, Connecticut, Kansas and Massachusetts were added to the list (Figure 1). Despite our best efforts at quarantine and the imposition of fines for moving potentially infested firewood, the spread of EAB has continued. EAB has been responsible for the destruction of tens of millions of ash trees in Michigan in addition to the tens of millions lost in Canada, the Midwest, and a several states along the Eastern part of the country.
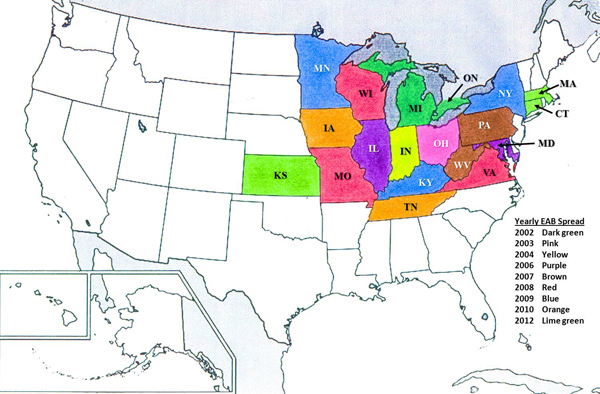
Figure 1: General schematic for the spread of EAB in the United States and Canada from the epicenter in Detroit, MI over the last decade.
While the focus in the United States and Canada has centered squarely on the elimination of EAB, the main threat to our ash trees, many colleagues in Europe have had another focus. Within the last decade an emerging infectious pathogen Hymenoscyphus pseudoalbidus has rapidly infected and killed millions of European ash trees. The initial symptoms of infection are small necrotic spots on stems and branches that enlarge to form perennial cankers, wilting, and crown death (Figure 2).
Prior to identification of the fungus, trees in Poland were reported to be dying in large numbers in the early 1990s followed by vast numbers of ash trees in Estonia, Latvia, and Lithuania. This disease was first described in 2003 in Denmark but it was not until 2006 that identification of the asexual stage, Chalara fraxinea was designated. Two years later, in 2008, scientists confirmed that the disease was caused by a fungus and in 2010 Hymenoscyphus pseudoalbidus, the sexual stage, was identified and coupled with the asexual stage. This breakthrough occurred after the infection had spread throughout Austria, the Czech Republic, Germany, Scandinavia, Slovenia, and Switzerland. At last count, Belgium, France Great Britain, Hungary, Ireland, Italy, the Netherlands, Romania and Russia (Figure 3) were all infected.

Figure 2: Symptoms of Hymenoscyphus pseudoalbidus infection are small necrotic spots on stems and branches that enlarge to form perennial cankers, wilting, and crown death.
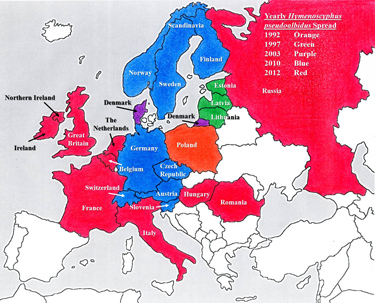
Figure 3: Hymenoscyphus pseudoalbidus has spread across the majority of European countries within the last decade.
Often a chronic infection, older trees can survive initial attacks before eventually dying but younger trees are killed within a single growing season. The rapid spread of the fungus over the last decade has discouraged many plantation owners and foresters from planting ash trees and the remaining trees are being quickly harvested before the lumber quality and lumber prices decline. Thus far, few ash trees have shown any resistance to the fungus and researchers at the University of Copenhagen have proposed that fewer than 5% of trees demonstrate resistance at levels high enough to survive fungal attack (McKinney et al. 2011), an attribute that may be due to a genetically-linked defense response (McKinney et al. 2012). Early estimates state that 90%–99% of the 80 million ash trees in Denmark will likely die. Researchers in Lithuania have identified 50 disease-resistant trees for use in breeding populations. Knowledge of heritable variations in susceptibility may be beneficial for eventual repopulation efforts but are unlikely to be helpful in the short-term.
Comparative analysis between our efforts to eradicate the EAB are mirrored in efforts of the European cohort with Hymenoscyphus pseudoalbidus as both groups have found that banning of importation and movement of ash wood (Ireland), removal of infected trees (Great Britain), and destruction of all trees within a certain area have all failed to slow the spread (Denmark). Innovative methods such as the creation of a phone application called “Ashtag” to track disease sightings have been employed to improve information flow. Furthermore, studies of Manchurian ash (Fraxinus mandschurica) trees have indicated, as for EAB, trees in Asia appear immune to the fungus while European ash (Fraxinus excelsior) and Narrow-leafed ash (Fraxinus angustifolia) are highly susceptible. Manna ash (Fraxinus ornus), another European species can also host the fungus but is less susceptible than many other European species. Evaluations of North American species have indicated that Black ash (Fraxinus nigra) and to a lesser extent Green ash (Fraxinus pennsylvanica) are susceptible while White ash (Fraxinus americana) trees appear highly resistant.
Efforts are being made in labs across Europe to identify resistant populations but the future of many European and North American ash trees is bleak. With strict regulatory procedures in place to prevent the generation and dispersal of transgenics (trees engineered to express foreign or modified DNA sequences) that may be resistant to the fungus or EAB, researchers are forced to consider more creative methods of retaliation. Use of biological controls such as parasitic wasps, natural predators (woodpeckers), and pathogenic fungi have been used in the United States with limited success against the EAB. These new findings highlight the urgent need for early screening and selection for fungus-, disease-, and EAB-resistant native populations for use in breeding studies. Removal of all ash trees within an area may eliminate trees with innate resistance as well as the infected. Continued ash breeding efforts may result in increased resistance to either the Hymenoscyphus pseudoalbidus fungus or EAB however a long-term time commitment must be made if we are to make progress towards saving one of our most valued hardwoods.
Cited References
McKinney LV, Nielson LR, Hansen JK, Kjӕer ED. (2011) Presence of natural genetic resistance in Fraxinus excelsior (Oleraceae) to Chalara fraxinea (Ascomycota): an emerging infectious disease. Heredity 106(5): 788–797. doi: 10.1038/hdy.2010.119
McKinney LV, Thomsen IM, Kjӕer ED, and Nielsen LR (2012) Genetic resistance to Hymenoscyphus pseudoalbidus limits fungal growth and symptom occurrence in Fraxinus excelsior. Forest Pathology 42 69–74. doi: 10.1111/j.1439-0329.2011.00725.x
Epicormic Branching in Oaks: Three Year Results
By Andy Meier – HEE Project Coordinator, Mike Saunders – Professor of Ecology and Natural Resources, Charles Michler– Director of HTIRC
In early 2010, we initiated a project to assess the impacts of silvicultural treatments on epicormic branching in oak, with a particular focus on white oak (see HTIRC newsletter, April 2010). Epicormic branches are small, new branches that form throughout the tree from buds that have been dormant for a long period of time. They are most evident and have the greatest impact on timber value when they form on the merchantable bole of a tree (Figure 1). Oaks in general, and white oak in particular, are known to be especially susceptible to loss of log grade due to epicormics.
Because epicormics are routinely observed following disturbances in the forest canopy, it has been thought that sudden increases in light stimulate epicormic sprouting. Other factors have also been implicated. Prior research has shown that low vigor trees tend to produce more epicormics than dominant, high vigor trees. At the same time, there can be noticeable variation among trees of similar vigor classes within an individual stand. This has led to the assumption that there is some level of heritable genetic control over epicormic branching. The main objective of our project was to determine the relative influences of genetic background and tree vigor on the tendency to form epicormics and how increased light associated with silvicultural treatments influences this tendency.
This project consisted of three studies, each corresponding to a different aspect of epicormic bud and branch development. For two of these we utilized trees from two white oak progeny tests planted from acorns collected in 1983 throughout Indiana, Illinois and Missouri (see HTIRC newsletter, January 2008). In spite of the assumption of substantial genetic variation in epicormic branch production, very little data has been published quantifying epicormics at either the stand (provenance) or family level, so these progeny tests allowed us the unique opportunity to quantitatively compare epicormics among families. Some trees from these progeny tests were subjected to release treatments that removed all competing trees (Figure 2) and the epicormic response over three years was studied on location. Other trees were harvested and brought back to Purdue University, where prior epicormic development was assessed through the use of computed tomography (CT) scanning (Figure 3). This was possible because the unique formations produced by epicormic buds and branches in the wood, known as epicormic traces, could be easily counted and measured in the resulting images (Figure 4). Our third study used sites on properties throughout Indiana (Figure 5) managed by Purdue University, the Indiana DNR – Division of Forestry and as part of the Hardwood Ecosystem Experiment, where timber harvests in the past 15 years had created large openings and, consequently, exposed trunks of trees along the borders of the openings to full sunlight. We counted the number of epicormics on the bottom two 16-foot logs of oaks adjacent to these openings and related these numbers to a wide array of tree and site variables.
Our findings for these three studies were not necessarily surprising though they were somewhat contrary to traditional thought. In the progeny tests, we found that the total number of epicormic branches were no different three years following release than they were prior; in fact, the mean number of branches per tree actually decreased slightly. There was, however, a significant increase in the size of epicormic branches for most trees. Trees with the most large branches prior to treatment also tended to have the most large branches following treatment. We did observe a small family effect on total branch size, but this family effect was relatively minor in comparison to the influence of pre-treatment branch characteristics. Rather than producing new branches, trees in this study appeared to be able to allocate more resources to increased development of epicormic branches already present.
In the unreleased trees subjected to CT scanning, the number of epicormic traces present was very strongly related to the total number of regular branch knots that we observed in scanned images. However, the proportion of these traces that showed an indication of development (i.e. sprouting) was generally much lower for trees with high growth rates. Variation within families in total epicormics was high, while there was little variation in means among families. The implication from this, therefore, is that trees with more regular branches produce more epicormic buds but epicormic buds on vigorous, rapidly growing trees are less likely to sprout.
Taken together, the two studies from the progeny tests show the major influence of prior branch development on epicormic responses to silvicultural treatments. Vigor seems to be an important component that influences bud sprouting, while genetic variation was of only minor importance. The genetic influence may be much more important in younger trees when epicormic buds are being established. Further study is needed to verify this hypothesis.
Finally, in the third study, tree diameter was by far the most important factor related to the number of epicormics on the bottom two sawlogs. At larger diameters (above about 15 in. diameter at breast height (dbh)), epicormic numbers were almost always low, regardless of oak species. Below 15 in. dbh, trees had more epicormics, and black oaks in this group had more epicormics than chestnut, northern red or white oaks. Above 21 in. dbh, trees on average had only 2 epicormics. We did not find strong variation in the total number of epicormics that was explained by any of the other factors. There was variation among sites, but much of this could be explained by differences in mean stand diameter. Sites with the lowest mean diameters had generally higher mean numbers of epicormics on the bottom two 16-foot logs, while sites with higher mean diameters had fewer epicormics. This again suggests that epicormic branch numbers are influenced more by biologically controlled developmental dynamics than by increases in light following silvicultural treatments.
In summary, from the past three years of work on epicormic branching in oaks we have been able to show that 1) epicormic branching following silvicultural treatments is a more complicated phenomenon than a simple response to light, 2) variation among families in epicormic tendencies is present but other factors such as tree vigor and previous epicormic development are probably more important and 3) epicormic prevalence following silvicultural treatment is strongly related to the number of epicormics present prior to treatment. From the perspective of tree improvement, these studies show that it may be possible to select against epicormic branching at an early age by selecting trees or families that have a lower number of regular branches. It is also clear that, to limit epicormic branching in stands of improved trees, silvicultural treatments must be implemented that maintain tree vigor. From a silvicultural perspective, our studies and others indicate that proper selection of crop trees should limit degrade from epicormics in oak stands; by retaining vigorous trees that have no epicormics the likelihood of new epicormic development is small. However, thorough examination of potential crop trees is necessary to ensure that there is no evidence of prior epicormic development.

Figure 1 Example of epicormic branches on the trunk of a mature white oak

Figure 2. Fully released white oak crown three years following treatment
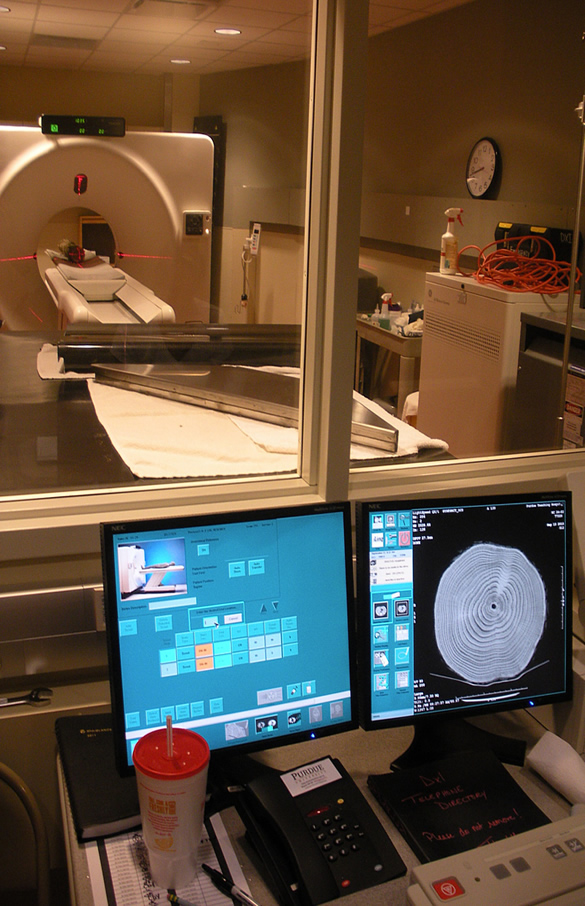
Figure 3. White oak log being scanned
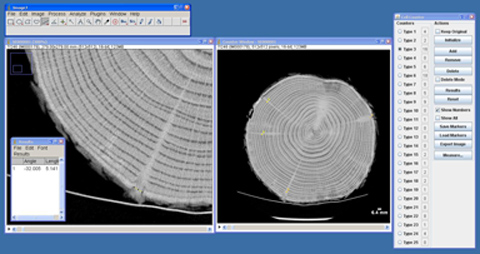
Figure 4. Analysis of scanned log image showing multiple epicormic traces
Indiana’s Maples May be Next on the Menu
By Annemarie Nagle, the Forest Pest Outreach Coordinator in the Dept. of Entomology at Purdue University
Annemarie Nagle
Many Indiana communities are suffering from, or preparing for, the impacts of the emerald ash borer (EAB) infestation, which will eventually cost the state over 147 million ash trees and millions of dollars in lost timber values and urban ash management. By the time the infestation encompasses the native range of Fraxinus, EAB will be the most destructive invasive forest pest in US history. While EAB has been nothing but bad news for Indiana’s forests, worse news may be coming. Asian longhorned beetle (ALB), EAB’s bigger, hungrier cousin, is lurking on our doorstep. The most recent infestation of ALB was found in 2011 in Bethel Ohio, a small rural community near Cincinnati and 40 miles from the Indiana state line.
In long-infested communities ALB is known as the “Darth Vader bug,” a commentary not only on its’ appearance but also on its destructive capacity. This potential invader threatens 19% of basal area nationally if it spreads out of control. As a comparison, EAB threatens 3%. ALB prefers maple trees (Acer), but is able to fatally infest 12 other native tree genera, including Aesculus (buckeyes and horsechestnuts), Ulmus (elms), Salix (willows), Betula (birches), Platanus (sycamores), Populus (poplars), Albizia (mimosa), Cerciphyllum (katsura), Fraxinus (ashes), Koelreuteria (goldenraintree), Sorbus (mountain ash), and Celtis (hackberry).
ALB is a wood-boring beetle native to China and Korea, where it has emerged as a major pest of elms, poplars, and willows, in plantations and windbreaks. Many of these plantations produce poor-quality lumber used to construct crates and pallets, which in turn are used to ship goods (and resident ALB larvae and adults) around the world. Though international trade is typically responsible for initial ALB introductions, movement of infested branches and logs from killed trees is often the source of new, satellite infestations.
In the US, ALB is a federally regulated pest, and eradication is the goal whenever it is found. Ohio is the fifth state to attempt eradication of this devastating tree killer, which has also been found in New York (Brooklyn, 1996), Illinois (Chicago, 1998), New Jersey (Jersey City, 2002), and Massachusetts (Worcester, 2007). Success is possible; ALB has been eradicated from several European countries, Chicago, parts of New York, and most recently, New Jersey (it took 11 years and destruction of over 20,000 trees).
In the US, eradication of ALB outbreaks has been achieved with control strategies involving extensive delimiting surveys, removal and destruction of infested trees, and chemical treatments or removals of trees in ½ mile buffer zones around known infestations. As one might imagine, both the beetle’s feeding damage and efforts to eradicate it have profound impacts on infested communities. To date, nearly 9,200 of Bethel’s deciduous trees have met the saw and chipper. Sixty-one square miles remain under strict quarantine, limiting movement of potentially infested tree materials, while survey crews still struggle to gain a handle on how far the infestation has spread. As is the case with most invasive species, early detection of ALB outbreaks is imperative to the success of eradication.
Fortunately, ALB has yet to be found in Indiana, but its’ entry through the firewood or international shipping pathways is of grave concern. Keeping a lookout for the ALB and its damage could mean the difference between life and death of trees in your town and on your property.The following ALB diagnostics, originally published as part of an article in The Woodland Steward will help you know what to look for.
ALB Diagnostics
Look for ALB damage on LIVING trunks and branches of its preferred host trees.
Holes or Round Pits in Tree Branches or Trunk
ALB creates very characteristic damage on the trunks and limbs of infested trees. During egg-laying, adult females chew oblong bowl-shaped pits into the tree’s bark. Egg-laying pits are typically about ½” diameter and weeping sap is sometimes present. Emergence holes are created when newly formed adults chew their way out from their larval home. These large, 3/8” to ½” holes (about the diameter of a #2 pencil) penetrate deep into the tree. Heavily infested trees are often riddled with egg-laying pits and emergence holes, and coarse sawdust may collect around the tree base or in branch crotches. Woodpeckers also create holes in infested trees to consume ALB larvae. Holes created by woodpecker feeding tend to be much wider than egg-laying pits, and extend deep into the wood of the tree.
Broken Branches with Large Larvae
The juvenile, or larval stage of ALB packs the tree-killing punch. Larvae are legless off-white grubs that may be visible in freshly cut wood. After hatching just beneath the bark, larvae feed only briefly on the tree’s nutrient-conducting tissue. Most feeding is in the structural wood of infested trees, creating ½” wide tunnels until the heartwood resembles Swiss cheese. The relatively minor damage to the tree’s vascular system allows it to stay green and appear healthy, making canopy dieback an unreliable diagnostic. Undetected infestations are extremely dangerous because they can fill the canopy with healthy-looking but structurally unsound limbs, which often snap off during storms and wind events.
Large Black and White Adult Beetles
All U.S. infestations have been identified by alert citizens reporting these unique and conspicuous beetles. ALB adults are large (1-1 ½” long) shiny black beetles with irregular white spots on their backs, sometimes with blue tinged feet. They are named for their long, black and white striped antennae, which exceed the length of their bodies. Beetles begin emerging from infested trees in late May, and continue through October. Adults are capable of flight, but typically do not move far from their parent tree, tending to re-infest it until the tree is dead.
Think You’ve Found ALB? Report It!
Never assume invasive pests are “somewhere else.” If you see any beetle that looks like ALB or any signs that resemble ALB damage, take a photo and send to Indiana DNR Division of Entomology and Plant Pathology at DEPP@dnr.in.gov. Capture and hold onto the insect for identification if you can. You can also call DNR’s Invasive Species Hotline at 1-866-NO EXOTIC (1-866-663-9684). To learn more about signs and symptoms visit www.asianlonghornedbeetle.com.

Photographer: Dr. Cliff Sadof, Purdue Entomology. A fully grown ALB larva.

Photographer: Dr. Cliff Sadof, Purdue Entomology. ALB Egg laying pits and exit holes
on an infested maple trunk.
The tree had a healthy-looking canopy despite heavy infestation.

Photographer: Annemare Nagle, Purdue Entomology. An adult ALB.
References:
Boggs, J. and Stone, A. “Asian Longhorned Beetle: The Threat in Black and White.” Tree Care Industry Magazine. April 2012. http://www.tcia.org/digital_magazine/tci-magazine/2012/04/index.html (accessed Jan. 15, 2013).
Hobson, D.A.; Tyrrell, M.L.; Camp, A.E., eds. “New Threats to North American Forests.” Yale Forest Forum Review. 2003. Vol. 6 (1). http://environment.yale.edu/gisf/files/pdfs/yff_reviews/02.03.pdf (accessed Jan. 15, 2013).
Nagle, Annemarie. “Your Maples—Next on the Menu?” The Woodland Steward. Spring 2013. Vol. 22 (1). http://woodlandsteward.squarespace.com/storage/past-issues/Web_Woodland_Steward-Spring_2013.pdf (accessed April 2, 2013).
From Zamorano, Honduras to West Lafayette, Indiana – HTIRC Contemplates a Dual-Language Initiative!
By Shaneka Lawson, Plant Physiologist
Typically when you think of reading the latest research results from the HTIRC group, you rarely think about whether or not those results will be in English. For students and colleagues in countries where the primary language is Spanish, reading scientific research results and gaining a thorough understanding of the work presented is often difficult. A new theory to help with this problem was proposed by Operation Breeder Jim McKenna. It seems likely that if students were able to both read and comprehend the research being done at Purdue University within the HTIRC and TropHTIRC, a greater number of Spanish-speaking students and researchers would be interested in the projects being investigated.
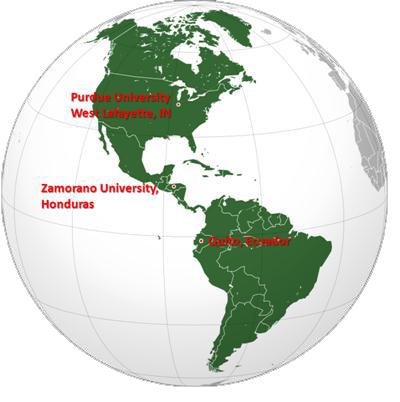
Figure 1. Locations of Ecuador and Zamorano University (Honduras) in relation to Purdue University.
After working with the latest visiting student from Zamorano University; I too agree that students in Spanish-speaking countries interested in coming to work at Purdue would be more informed with a bi-lingual presentation of research data. It is probable that visiting students would have a more vested interest in a research program that makes an attempt to overcome the language barrier to offer opportunities to learn aspects of forestry within the laboratory and out in the field.
The most recent visitor from Zamorano University is Miss Karen Hidalgo, a 21-year-old student from Ecuador interested in both urban forestry and tree physiology (Figure 2). Karen will be the first student to write a synopsis of her research experiences during her four month visit in both English and Spanish. Upon her return to Ecuador the research goals and photos of her experiences will be used in a presentation to other students about the objectives of the visiting scholar program and the research opportunities within the HTIRC. This initiative is just one of the many ways that the HTIRC is breaking new ground.
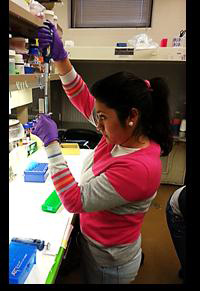
Figure 2: HTIRC visiting student Karen Hidalgo works to isolate DNA in the lab.
Thousand Cankers Disease Webinars Provide New Information for 2013
On March 28 over 250 people from 26 states and Canada participated in a live online webinar on Thousand Cankers Disease (TCD). Manfred Mielke of the US Forest Service Forest Health Protection moderated separate presentations on the walnut twig beetle, the Geosmithia morbida fungus, 2012 national survey results, and APHIS guidelines. In addition, state personnel from Pennsylvania, Virginia, Tennessee, North Carolina, and Ohio gave updates on the status of the disease in their states and past and future actions to address this disease complex.
This webinar was presented by the Hardwood Tree Improvement & Regeneration Center at Purdue University, USDA Forest Service State & Private Forestry and Forest Health Protection, Purdue University Department of Entomology, and the Walnut Council. Audience members included landowners, natural resource professionals, state agency personnel, master gardeners, and forest scientists.
Attendees were able to submit questions via text as they listened to the speakers. Of clear interest to a number of questioners was the progression and length of the disease in a specific tree or stand. Although several speakers discussed that question, the consensus was that the science and experience is still too new to be certain, but many infestations seem to take 5-20 years from initial insect to tree mortality.
The next webinar is scheduled for April 25 on the topics of 2013 state surveys and implementation, field identification of TCD, and a FHTET (Forest Health Technology Enterprise Team) demo on survey database entry.
The May 30 webinar will provide an update on current research and methods and public outreach plans and needs.
Registration is not necessary to attend future webinars. Go to
http://www.thousandcankers.com/webinars.php for more information on future meetings or to view recordings of past webinars.
Have questions about tree planting? This series of publications can be viewed or downloaded free of charge. Planting and Care of Fine Hardwood Seedlings
Van Eck Scholarships available for graduate research with the HTIRC.
Ask the HTIRC: email Lenny Farlee with your tree planting and forest management questions and we’ll help you find the answers.
