HTIRC E-NEWSLETTER – July 2015
Volume 8 Issue 1
Articles in this issue:
- Manfred Mielke is Acting Director of HTIRC
- Use of Plastic Bottles as Nursery Containers for Growing Forest Tree Seedlings
- Advancements in Black Walnut Propagation
- Development of North American Ash Trees Expressing a Bacillus thuringiensis (Bt) Protein for Management of the Emerald Ash Borer
- Recent Proceedings Article Summarizes 10 year Performance of Select Walnut
- HTIRC Researcher Investigates the Use of Prescribed Fire in Hardwood Forests
- Where are Trees Going?
- HTIRC “On the Road” at Winter Events
- HTIRC Faculty, Staff, and Students Receive Awards and Honors
- Graduate School Opportunities with HTIRC
Location, Locations, Location – Using Stable Isotopes to View Site Health in Acacia koa Forests
Welcome to this issue of the Hardwood Tree Improvement and Regeneration Center E-newsletter. The HTIRC is committed to enhancing the productivity and quality of Central Hardwood Region trees and forests for the economic and environmental benefits they provide. Scientists at the HTIRC are using conventional tree improvement breeding as well as molecular and genetic technologies to improve the wood quality, growth characteristics, and insect and disease resistance of trees like black walnut, black cherry, red and white oaks, butternut and American chestnut. Research in tissue culture, tree nursery practices, tree plantation establishment and management, and Central Hardwoods silvicultural systems is aimed at increasing the regeneration success rate for high quality hardwood trees and forests. Some interesting and unusual research areas include examining the potential for propagating trees with “figured” wood: birds-eye maple or curly walnut; and breeding trees that will be an economical source of bio-fuels. Twice per year we will attempt to provide interesting and useful information on Central Hardwood trees and forests, as well as sources for additional information and assistance. Please pass this newsletter along to others who may enjoy or benefit from the information provided. If you would like a closer look at the HTIRC, please visit our web site at: http://www.htirc.org
Manfred Mielke is Acting Director for HTIRC

Manfred Mielke has been appointed Acting Director of HTIRC as Charles Michler recovers from health issues. Manfred has had a long and successful career as a Plant Pathologist for the USDA Forest Service, Forest Health Protection. He has cooperated with Northern Research Station, University scientists and HTIRC over the past 35 years on butternut canker, black cherry improvement, Thousand Cankers Disease, oak wilt, beech bark disease, sudden oak death and other native and introduced forest diseases. He was the long-time chair of the Walnut Council Protection Committee, past President of the National Walnut Council, and current President of the Wisconsin Chapter. He plans to continue the tradition of leadership by the Forest Service, University, and industry in the development of improved fine hardwoods and the restoration of threatened trees.
Use of Plastic Bottles as Nursery Containers for Growing Forest Tree Seedlings
By Safiullah Khurram, MS Graduate Student (Recently Graduated), Purdue University, Owen Burney, Assistant Professor, New Mexico State University, and Douglass Jacobs, Fred M. van Eck Professor, Purdue University
Deforestation and forest degradation are major global issues, especially in poor and developing regions of the world. To combat deforestation, it is critical to enhance the productivity of forest regeneration and restoration operations. Production of low quality seedlings with deformed and spiraled root systems hampers restoration programs in many developing countries. Due to resource limitations and a lack of modern technology, polybags (small plastic bags) are a common container type for plant seedlings propagation in these countries. However, polybags often produce seedlings with spiraled and deformed root systems. When seedling roots encounter the smooth wall of polybag, they begin to grow horizontally and circle around the root system (Figure 1). Planting seedlings with spiraled roots can result in poor establishment success as spiraled roots may girdle the entire root system and prevent new root initiation and absorption of water and nutrients (Watson and Himelick 1997).
Use of discarded plastic beverage bottles could be a viable alternative as nursery containers for propagation of quality seedlings in restoration programs. In 2009, approximately 120 billion plastic water bottles were used worldwide and it was estimated that another 120 billion were consumed for carbonated soda drinks (Gleick 2010). These bottles are used briefly, usually only for the duration of consumption from a single user after which the bottle is discarded. Where recycling and waste management are limited, these bottles end up in streets, waterways, and open areas. Generally, use of these discarded plastic bottles as growing containers has the two-fold advantage of reducing waste and extending the life of these products for an important cause. Likewise, use of plastic bottles would reduce the consumption of plastic in agriculture. The United States used 521 million pounds of plastic in agriculture during 1992, of which 66% was used for nursery containers (Amidon 1994). In 2002, approximately 1.7 billion pounds of plastic was used in the agriculture sector (Levitan and Barros, 2003). The texture, pattern, color, and thickness of these plastic bottles vary greatly, and these properties may influence growing conditions for individual seedlings. Thus, prior to using them as alternative containers, research is required to examine seedling performance under varying bottle properties.
We initiated a research project to assess the alternative option of seedling production using repurposed plastic bottles for growing quality native trees to benefit agroforestry, reforestation, restoration, and conservation programs in developing countries. The bottles used in this study were typical 0.5 L bottles obtained from the Coca-Cola Bottling Company. Two separate experiments address the overall study objective. In each study, we modified the plastic bottles by first removing the upper part of the bottle at the taper followed by placing six drainage holes at the bottom (Figure 2). The first experiment compared seedling performance of two different tree species, Arizona walnut (Juglans major (Torr.) A. Heller) and Afghan pine (Pinus eldarica), grown in bottle containers, polybags, and modern container types (Figure 3). In summary the results showed that bottle containers produced seedlings with similar morphology and root system architecture as the modern container type (DeepotTM), while polybags produced a large number of spiraled roots.
In the second experiment, the effects of bottle opacity and root spiraling control techniques were evaluated on seedling root system architecture and morphology. Root spiraling control treatments were based on air-slits (three vertical), interior ridges (three vertical), or a control with no root spiraling control technique. Opacity treatments were based on three bottle colors: black, clear and green (Figure 4). Results showed that opacity did not have a significant impact on seedling growth or root system architecture. However, both root spiraling prevention techniques (air-slits and inside-ridges) resulted in significant prevention of root spiraling compared to the control treatment.
The outcome of our project will help to educate our target audience of nursery production managers in developing countries on how to appropriately modify and use discarded water and beverage bottles as nursery containers for seedling propagation. Additionally, this project will help to produce high quality seedlings and enhance the productivity of restoration activities. In the future we hope to increase awareness of the advantages of using plastic bottles over polybag systems to produce quality seedlings for reforestation and restoration efforts in developing countries and worldwide
Note: For further information you can look to a Purdue University published thesis
Khurram, Safiullah. M.S., Purdue University, May 2015. Use of Plastic Bottles as an Alternative Container Type for Propagation of Forest Tree Seedlings in Restoration Programs. M.S. Thesis, Purdue University. 115p.

Figure1. Root spiraling (circling) around the tap root system, typical
for seedlings
grown in smooth walled polybag container type.
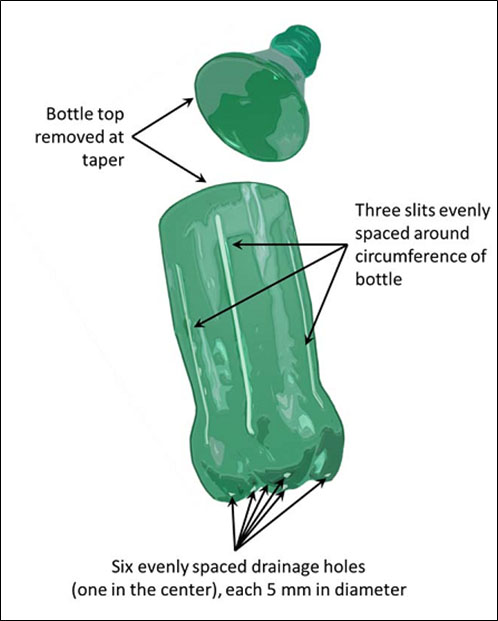
Figure 2. Bottle modification provided for nursery grown container

Figure 3. Comparison of bottle containers (A and B) with polybag (C) and modern container DeepotTM (D).

Figure 4. Comparison of three bottle opacities (clear, green, and black; left to right)
and three spiral control techniques (control, interior-ridges, and side-slits; left to right).
References:
Amidon. 1994. Use and disposal of plastics in agriculture. A report prepared by Amidon Recycling for the American Plastics Council.
Gleick, P. H. 2010. Bottled and sold: The story behind our obsession with bottled water. Washington, DC: Island Press.
Levitan, L. and Barros, A. 2003. Recycling Agricultural Plastics in New York State.‖ A research report prepared for the Environmental Risk Analysis Program, Cornell University, Ithaca New York.
Watson, G. W., and Himelick, E. B. 1997. Principles and practice of planting trees and shrubs. (Pp. 107-113). Champaigne, IL: International Society of Arboriculture.
Advancements in Black Walnut Propagation
By Micah E. Stevens, PhD Graduate Student and Dr. Paula M. Pijut, Plant Physiologist
A commercialized, clonal black walnut propagation system not reliant on the time and labor intensive grafting process remains elusive, but recent strides have been made with in vitro and softwood cutting propagation. Current research at Purdue University and the Hardwood Tree Improvement and Regeneration Center (HTIRC) in West Lafayette, IN has sought to not only improve propagation, but also to better understand why black walnut rarely produces adventitious roots. Adventitious roots are defined as roots that form on any aerial portion of a plant, and the inability to form these roots is common in several woody perennials such as black walnut. With an eye to eventual commercialization, the questions we are investigating will hopefully lead to techniques that allow more efficient and cost effective black walnut clonal propagation.
In vitro micropropagation allows a large volume of genetically identical material to be produced in a relatively short period of time. We have been using elite black walnut genotypes selected by the HTIRC for our in vitro work, and have successfully developed a system by which plant material can be collected from greenhouse grown trees and thrive under aseptic conditions (Fig. 1A) and rapidly multiply (Fig. 1B). This novel system for black walnut shoot proliferation, which significantly increased the rate of shoot multiplication compared to established techniques, will allow us to further study adventitious root formation in black walnut. These results will allow us to further study the complex molecular and physiological process of adventitious root formation, a major hurdle in clonal walnut propagation.
Apart from developing rapid shoot multiplication of in vitro material, we have also been able to successfully root black walnut softwood cuttings. After excision from the “mother” plant, softwood cuttings are extremely sensitive to humidity and can quickly dry out and perish if not maintained under high humidity. This increase in humidity allows the plant cuttings an opportunity to thrive in order to form roots and continue to grow. Past attempts to root walnut softwood cuttings required significant pre-treatment that would make commercial application impractical. By using a fog system to maintain an elevated humidity and plant growth regulators to induce rooting, walnut cuttings were successfully rooted as high as 71% with our method. The use of fog to root cuttings is commonplace in many horticulture systems, but has rarely been applied routinely to forest tree species. Using our method, rooted cuttings were healthy with well-developed root systems (Fig. 2A and B), and continued to grow normally after transplanting to soil (Fig. 2C). Our research continues to improve clonal black walnut production as well as to better understand some of the major roadblocks preventing routine propagation. By employing these novel techniques we have begun to improve black walnut propagation, a perennially difficult species to study. Continued efforts are especially urgent given the recent detection of Thousand Cankers Disease in the natural range of black walnut. Efficient and reliable propagation methods, such as the ones we are developing, are powerful tools for tree breeders and for conservation efforts.

Figure 1. In vitro grown black walnut (Juglans nigra)shoots. A) Elongated shoots initiated from
a single node collected from greenhouse grown tree. B) A mass of aseptically
grown black walnut shoots rapidly multiplying.

Figure 2. Successfully rooted black walnut (Juglans nigra) softwood cuttings.
A) A healthy rooted cutting 5 weeks after adventitious root initiation. B) A detailed look
at the healthy root system of a rooted cutting. C) A cutting that has been rooted
and transplanted to soil and continuing to grow normally.
Development of North American Ash Trees Expressing a Bacillus thuringiensis (Bt) Protein for Management of the Emerald Ash Borer
Dr. Paula M. Pijut, Research Plant Physiologist with the HTIRC, would like to acknowledge and thank several contributors to her Ash (Fraxinus spp.) – Emerald Ash Borer research program. During the April 2015 Board meeting of the National Hardwood Lumber Association (NHLA) the following companies committed funding support to this research program. Special thanks to members of the NHLA Board of Directors: Pike Lumber Company Incorporated, Cole Hardwood Incorporated, Atlanta Hardwood Corporation, Northland Corporation, J.T. Shannon Lumber Company, Rossi Group, and Turn Bull Lumber Company. Dr. Pijut would also like to acknowledge and thank the U.S. Endowment for Forestry and Communities for their support of this project through a research grant.
Recent Proceedings Article Summarizes 10 year Performance of Select Walnut
An article in the National Proceedings: Forest and Conservation Nursery Associations – 2013 – RMRS-P-72 by James R McKenna, Operational Tree Breeder, HTIRC, and Philip A O’Connor, Tree Improvement Specialist, Indiana DNR Division of Forestry, examines the performance of seven populations of black walnut seedlings planted in 2002 at two locations in Indiana. Two populations were from orchards of trees selected for superior form and growth at Purdue University and Indiana Division of Forestry properties, one population was a wild-type forest population, and four others represented typical collection sites for walnut seed from plantations, yards, parks and similar locations. In addition, one population was seed from an open pollinated black walnut clone (termed a half-sib family since the mother but not the father tree is known) selected for superior traits and representing a population of low genetic diversity.
The two planting sites were in southern (Martinsville) and central (Fishers) Indiana. The soil at Fishers is an Ockley silt loam, fairly uniform, with a slight slope toward a creek. The NRCS Black Walnut Suitability Index (BWSI) characterizes this soil type as “well suited for walnut.” At Martinsville, two soil types occur: a Wilbur silt loam that the BWSI characterizes as “moderately well suited” for black walnut, and an Ava silt loam that the BWSI characterizes as “somewhat suited” for walnut. Unlike the Fishers site, the soil is quite variable with gravel and clay deposits in places, and neighboring hillsides that cause intermittent flooding during heavy rainfall events throughout the year. At Martinsville, trees grew dramatically different between the two soil types. Six blocks of the bulked populations that were planted on the Ava silt loam were ultimately discarded from the analysis as the soil proved unsuitable for walnut, leaving four blocks grown on the Wilbur soil to analyze. The half-sib family was planted on the Ava soil and the poorest half of those seedlings was discarded from the analysis.
Three years after planting, survival, height, and presence or absence of a single-leader differed significantly. At Martinsville, survival averaged 84 percent overall with individual populations ranging from 72 to 91 percent. Survival averaged 98 percent at Fishers, with populations ranging from 94 to 100 percent. The SEPAC population had the lowest survival at both sites. Tip-dieback and stem cankers affected 57 percent of seedlings across all populations at Martinsville compared to just 6 percent at Fishers. These diseases were primarily due to a large infestation of ambrosia beetles, particularly at Martinsville, in 2004. The stem cankers appeared to be a result of either Fusarium or Phomopsis fungi, both of which cause annual cankers on walnut, but no known pathogen was confirmed. By the fifth year, survival remained unchanged at either site but growth varied significantly between sites. Trees were larger and more uniform at Fishers. Trees that had been affected by stem cankers at Martinsville had mostly recovered or re-sprouted from disease/insect problems encountered in years two and three. At Martinsville however, blocks planted on the Ava silt loam soil failed to thrive. Most of the half-sib family planted on this soil began to show signs of stress by five years. By the tenth year, quality remained significantly different among populations (table 2) while height and DBH differences still present were no longer statistically significant. The half-sib family performed very well at Fishers and had remarkable consistency in growth indicated by the low standard deviations for height and DBH. However, at Martinsville, on the Ava soil, the same family grew far below average. Under these poor growing conditions, the half-sib family still had good stem form although the percentage of trees with single leaders was greatly reduced.
Seedlings from the Purdue University clone bank, the Vallonia Subline 1 seed orchard, and from the half-sib family, all had significantly better stem quality than did the other five bulked populations throughout the study. Increasing the single leader percentage should minimize the need for pruning co-dominant leaders and increase the length of merchantable logs. Raising the stem quality rating should reduce the need and volume of corrective pruning along with elevating the timber-grade of trees, which ultimately will increase the timber value. Both the Vallonia orchard and Martell clone bank material showed height growth trending above the plantation mean, but the results were not statistically significant. We found a similar trend with stem diameter (DBH), but again, by ten years, the differences were statistically insignificant suggesting that non-genetic factors have a large effect on walnut growth rates. It is not uncommon to find more genetic variation within populations than among them (Paul Bloese, personal communication 2013).
The performance of the half-sib family between the two different sites in the current study is very dramatic and highlights this difficulty. While the half-sib family had the highest overall improvement value at Fishers (+19 percent), it performed far below average at Martinsville. It was, however, inadvertently planted on a very poor soil at Martinsville, and as such is not directly comparable to the bulk populations grown there on the better soil. Clearly, utilizing too few test sites, and not controlling or accounting for soil differences within a given test site, can lead to erroneous conclusions on the genetic growth potential of walnut. This result underscores the extreme site sensitivity of walnut, and furthermore, the fact that genetically improved walnut will not show any gain when grown on an unsuitable walnut site. As we begin to make second cycle selections from a broad series of progeny tests, we expect to be better able to select for vigor by selecting larger individuals grown in even-aged plantations that already have an improved level of genetic quality for timber form. For the grower, a reduction in the number of slower growing and poorly formed trees, like the Vallonia Subline 1 and half-sib trees offer (figure 1), functionally improves the plantation. While improvement can be made culturally through pruning, staking, and fertilizing trees, such cultural management is costly and uncommon in forestry. Thus, having planting stock with genetic qualities for good timber form and uniform growth are very desirable. By utilizing select genetic stock that produces a higher percentage of acceptable trees, and grows more uniformly overall, pruning can be simplified and reduced, thinning decisions can become more systematic, and fewer walnut trees need to be planted which can make mixed hardwood plantings more successful (McKenna and Farlee 2013).
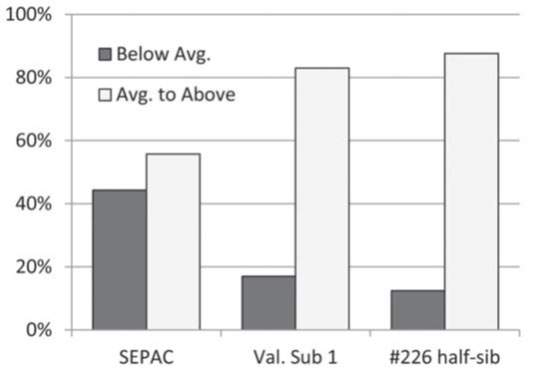
Figure 1. Proportion of 10-year-old trees rated below average or average to above average in quality of three populations. The Vallonia Subline 1 and #226 half-sib represent selections from parents with what appear to be superior characteristics, while the SEPAC population is from an average quality plantation. Below average trees scored a 1 or 2 stem-form rating at ten years and are candidates for early thinning. Average, above average, and excellent trees have a stem-form rating of 3, 4, or 5, respectively, and represent potential crop trees. Data from the Fishers site only.
Through progeny testing, tree improvement provides a means of weeding out poor performing seed sources as well as developing sources that will be bigger and better. Thus, source-identified and progeny tested seed, given good site selection and management, provides a grower confidence that trees have the genetic capacity to grow well. The practical reality is that walnut seed for reforestation will only rarely ever come from forest stands. By creating grafted walnut seed orchards from wild plus-trees we are able to capture genotypes from the forest, and by planting them together in an orchard, seed production and harvesting can be more economically managed. As replicated progeny test data becomes available, poorer performing clones can be eliminated from the Vallonia orchard, thereby converting it into an improved walnut seed orchard.
Some key points:
Management of high quality walnut has a much better chance of success on sites well-suited to walnut growth. High quality planting stock and management does not make up for poor planting sites in terms of tree growth and quality.
Screening tree families in a tree breeding program can help eliminate poorly performing families from the planting pool, as well as identify strong performers, providing more consistency and predictability, and perhaps reducing management costs.
You may view the full article at: http://www.fs.fed.us/rm/pubs/rmrs_p072/rmrs_p072_006_001.pdf
Citations
Bloese, P. 2013. Personal communication. East Lansing, MI: Tree Improvement Supervisor, Forestry Department, Michigan State University, 126 Natural Resources Building
HTIRC Researcher Investigates the Use of Prescribed Fire in Hardwood Forests
Michael Saunders, Associate Professor of Forestry at Purdue University and a Silviculturist with the HTIRC was recognized in a December, 2014 article in the Purdue Today web publication for his work on prescribed burning on 80 acres of the 63,000-acre oak forest at Naval Support Installation Crane in southwest Indiana.
There also will be burns in the spring and fall of 2015 and in the spring of 2016 in the Yellowwood and Morgan Monroe State Forests in the Bloomington area under a grant from the Indiana Division of Forestry. Those burns will be conducted as part of the Hardwood Ecosystem Experiment, a long-term study of forest management in Indiana and its effects on plants and animals. All told, there will be burns in about 200 acres at more than 15 sites.
Researchers want to determine how the prescribed burns will affect oak regeneration; wildlife such as salamanders and birds; and insects such as butterflies, spiders and beetles. “What we are doing is renewing the forest,” Saunders said. “The burns will increase the resiliency of the forests, increase oak regeneration and provide better wildlife habitat.”
Shade from the understory and midstory trees prevent oak seedlings from growing into mature trees. Saunders said that as older oak trees die in forests with heavy shade, there are no younger oak trees to replace them. Instead, the midstory trees eventually overtake the oaks. “If we don’t kill the midstory trees, then they eventually will become the overstory, which will change the forest,” Saunders said. Oak-dominated forests, which are exceedingly valuable to wildlife and wood products, could be replaced by maple-dominated forests throughout a large portion of the state.
Oak trees survive the burning because they are well adapted to fire. “They are much more able to respond after a fire than most other species,” Saunders said.
You can access the complete article at http://www.purdue.edu/newsroom/releases/2014/Q4/purdue-researcher-leading-burn-projects-to-regenerate-oak-forests.html
Writer: Keith Robinson, 765-494-2722, robins89@purdue.edu
Source: Michael Saunders, 765-430-1440, msaunder@purdue.edu

A prescribed fire moves through the oak forest at Naval Support Installation Crane in Indiana. (Purdue Department of Forestry and Natural Resources photo/Michael Saunders)

Purdue forestry and natural resources and HTIRC graduate student Ethan Belair sets a fire line with a drip torch to start of a prescribed fire at Naval Support Installation Crane in Indiana. (Purdue Department of Forestry and Natural Resources photo/Michael Saunders)

A prescribed fire has consumed much of the litter and small twigs in part of an oak forest at Naval Support Installation Crane in Indiana, leaving larger pieces of dead wood intact. (Purdue Department of Forestry and Natural Resources photo/Michael Saunders)
Where Are Trees Going?
By Johanna Desprez, MS Graduate Student (Recently Graduated)
Climate change isn’t affecting tree movement the way most research would suggest.
Climate change is expected to have wide spread impacts on the United States, including redistribution of tree species ranges. The temperature and precipitation patterns in the eastern U.S. have changed over the last 30 years, and because of these changes, we expect to see a majority of species moving north, as temperatures warm and precipitation patterns change.
However, we found only about 60% of species were shifting their abundances north and only 11% moved significantly north (Figure 2). The more prevalent trend was that 80% of species were shifting their abundances west, of which half moved significantly west. This westward trend is apparent in all ecoregions within our study.
While we expected that species would migrate in response to climate change, most studies investigating range shift in the U.S. do not find change. For that reason we chose to conduct a study that investigated if species abundances are shifting within their current range, as opposed to looking for species range shift. We used Forest Inventory and Analysis (FIA) Data collected by the U.S. Forest Service to calculate the mean center of species abundance from the 1980’s and 2011 for 98 species and genera. When we compare the two time periods, we can understand the direction in which species abundances have shifted over the last 30 years.
Climate change can modify the dynamics within a habitat. Rising temperatures can make some habitats more suitable, especially further north and at higher elevations. Changes in precipitation can also influence a habitat’s suitability by making water more or less readily available for trees. Given these changes in habitat, we expect that species ranges will change as the climate continues to change.
In the majority of the eastern U.S., the mean annual temperature has not changed substantially, but in the northern Midwest (MN, WI and MI) and parts of the New England there has been more warming (Figure 2A). In contrast, the majority of the eastern U.S. total annual precipitation has either not changed or increased, the southern coast is the exception. The central Midwest was where we have found an increase in precipitation by as much as 300 mm (Figure 2B).
We suspect that the majority of species are shifting west because of the increased precipitation and fire suppression in the western part of the study range. We found that generally the species that moved significantly west had higher precipitation tolerance compared to those that did not shift significantly west because more precipitation can be beneficial for new recruitment. Fire suppression could be contributing because since post-European colonization there has been a decrease in fire frequency and severity in the eastern U.S which has led to tree species moving into prairies that were previously stifled by fire.
In current climate conditions in the eastern U.S., precipitation seems to be more influential on species shifts than is temperature. However, as the global temperature continues to rise and rising temperatures in U.S. become more wide spread, temperature may become more influential, given that the ecoregion (Northern Hardwood Region) that had the most warming also had the highest percent (80%) of species shifting their abundances north (Figure 2).
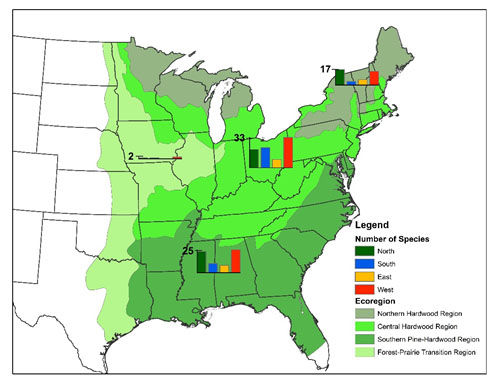
Figure 1- The number of species that shifted by ecoregion in each direction. Species abundances were collected by the Forest Inventory and Analysis (FIA) program.
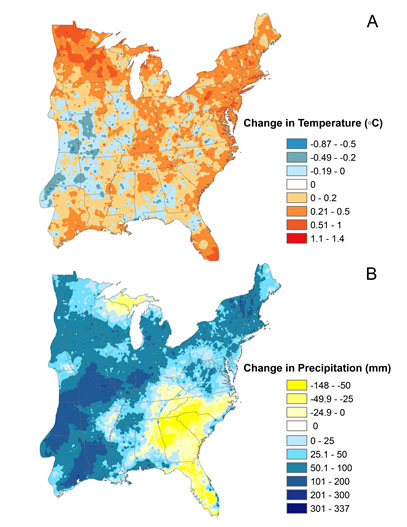
Figure 2- The changes in climate variables between the average of 1951-1980 and 1981-2010 (
PRISM Climate Group, Oregon State University, http://prism.oregonstate.edu, created 27 April 2015.) A) The change in annual mean temperature. B) The annual mean total precipitation.
Location, Location, Location – Using Stable Isotopes to
View Site Health in Acacia koa Forests
By Shaneka Lawson, Research Plant Physiologist
Isotopes that do not decay but persist in the environment are known as stable and can be used as indicators of forest health. The use of stable isotopes of carbon, hydrogen, nitrogen, and oxygen in studies of physiological processes within nature has been done for decades and continue to increase in popularity. In general, plants absorb a large volume of atmospheric CO2 (CO2atm) for use in photosynthesis. The breakdown of CO2 within the plant yields a ratio of carbon isotopes (δ13C:δ12C).
This article is focused on the use of stable carbon and nitrogen foliar isotopes, δ13C and δ15N respectively, in an analysis of Acacia koa forest nitrogen status and carbon dynamics. It has long been noted that isotope ratios vary depending on a number of factors from altitude, precipitation rate, season, and temperature. Previous work has shown that samples collected in winter months have somewhat depleted isotope values while summer values are generally enriched. This observation alone emphasizes the importance of seasonality in sample collection.
Isotopes of an element such as carbon are chemically the same or have the same number of electrons (carbon has 6 electrons) but have different physical properties because they differ in neutron number (proton number is the same) and atomic mass. These same variations in neutron numbers and atomic mass are seen in isotopes of nitrogen (7 electrons), oxygen (8 electrons), and hydrogen (1 electron).
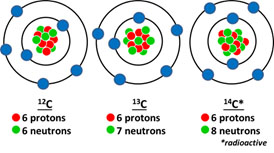
Isotope values are most often presented in the ‘delta’ value (δ) form that was presented above. These values are indicative of numerical ratios that convey the comparison of compositions between the sample and the standard accepted isotopic values. Delta values can be heavier (enriched) or lighter (depleted) than the standard. The majority of plant carbon is the standard δ12C while a much smaller fraction contributes to δ13C. The absolute abundance of isotopes is most often presented in the unit term of atom percent in scientific literature and in illustrative graphs. Atom percent is calculated as 13C = [13C/(12C + 13C)]*100 and is represented using ‰. The standard ratios of carbon isotopes (13C:12C) in C3 plants (most temperate crops, all woody trees), C4 plants (corn, sugarcane, and most grasses), and CAM plants (cacti, some orchid species) vary dramatically. The C3 plant ratio is usually between -33 to -24 ‰, C4 between -16 to -10 ‰, and CAM between -20 to -10 ‰ (O’Leary 1988).
Here is an example using carbon:
Foliar carbon samples were collected at the end of summer from a number of sites on the island of Hawai’i. Each site varied in elevation and average yearly precipitation. Samples collected from site #1 had an average δ13C value of (-31.89 ± 1.2‰) while the sample average from site #2 is (-26.22 ± 1.6‰).
What do these data tell us?
First, we know that several environmental conditions affect δ13C values such as CO2atm, irradiance (light intensity), and water status. Second, we know these data are from C3 plants, in this case Acacia koa trees. Third, we can see that the trees at site #2 have a greater (less negative) δ13C value. Research indicates that having a less negative δ13C means the plant has likely adapted to a drier environment and has a greater rate of water-use efficiency. Trees at site #1 are likely adapted to a wetter environment and therefore have lower water-use efficiencies because water is not as limiting at the location where those samples were collected.
Foliar samples from a number of our research sites in Hawai’i were accumulated and subjected to carbon and nitrogen isotope analysis to look for site-specific anomalies. While we have not concluded our examination of the collected data for patterns, we have noted trends and a few surprising irregularities in our data related to temperature, precipitation, and elevation. It appears at this point that several sites likely have soil that is more enriched in nutrients than other areas or that the unique microclimates at those sites have allowed koa trees to amass greater foliar carbon and nitrogen contents than similar sites.

Young Acacia koa
In the figure below (#10 with circle, arrows), a low elevation dry site shows a positive correlation (R2 = 0.46; blue line) between total foliar nitrogen (%N) and the stable nitrogen isotope (δ15N) but neither the mid- nor high elevation sites showed this relationship. Were we showing these data in addition to elevation, we would expect to see a negative correlation between elevation to stable nitrogen and elevation to foliar nitrogen as precipitation and nutrient availabilities decrease with an increase in elevation. This is just one of several unique findings from the sites selected for study.
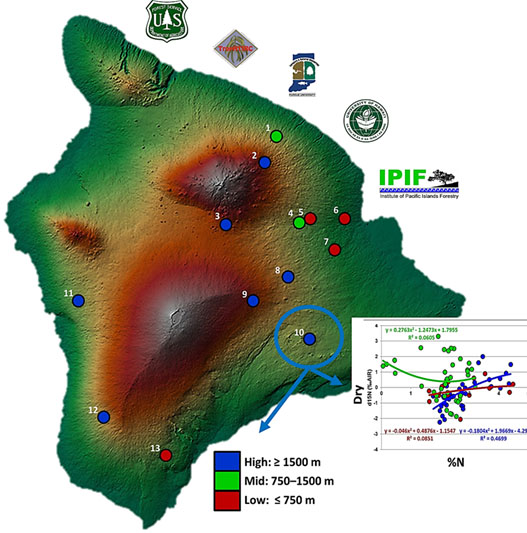
Stable isotope analysis is a popular method used to determine carbon fixation patterns, water-use efficiency, and an ever increasing number of additional applications in plant ecology and physiology. This sample data indicated that topographic variations as well as site conditions are important influences on nutrient cycling in koa forests.
Stay tuned for the final updates this fall as the manuscript pertaining to this research heads to press!
Reference:
O’Leary, M H. (1988) “Carbon Isotopes in Photosynthesis”. BioScience 38(5): 328–336. DOI:10.2307/1310735
HTIRC “On the Road” at Winter Events
An important part of our work at HTIRC is to share our research with the public, especially with the hardwood industry and landowners in the Midwest. This winter we went on the road to tell our story, to the Purdue University College of Agriculture annual Fish Fry and to the Indiana Hardwood Lumbermen’s Association annual convention.
Liz Jackson, engagement specialist, shared copies of the latest publications from the “Planting and Care of Fine Hardwoods” series, showed sample of walnut twig beetles and thousand cankers disease damage, and answered questions about forestry and HTIRC research. We appreciate the opportunity to get out in the public and learn more about what is important to forestry professionals and landowners and look forward to future opportunities to talk about our work. Our next event will be the national Walnut Council meeting in mid-June in St. Charles, Missouri.

Purdue Forestry & Natural Resources Alumni Coordinator Shelly Opperman and
HTIRC Engagement Specialist Liz Jackson met with attendees at the Purdue College of Agriculture fish fry.
HTIRC Faculty, Staff, and Students Receive Awards and Honors
Shaneka Lawson, Adjunct Assistant Professor, received the Multicultural Achievement Award from the USDA Forest Service.
Micah Stevens, Advisor Paula Pijut, received the Charles H. Michler Scholarship for the Outstanding Graduate Student in Forest Biology.
Teresa Clark, Advisor Songlin Fei, won First Place in the M.S./Research poster session at the 2015 FNR Research Symposium.
Kyle Earnshaw, Advisor Douglass Jacobs, and Charlotte Freeman won the Fischer Forestry Fund Graduate Student Scholarships.
Janis Gosewehr, Operation Assistant and Secretary, celebrated her 25th anniversary at Purdue this year.
Graduate School Opportunities with HTIRC
Hardwood Tree Improvement and Regeneration Center
Department of Forestry and Natural Resources, Purdue University
Description: The Hardwood Tree Improvement and Regeneration Center (HTIRC) at Purdue University is seeking outstanding candidates interested in working toward M.S. or Ph.D. degrees. Areas of research include: development of plant tissue culture and genetic modification technologies for tree improvement; conservation and population genetics; forest tree breeding; forest entomology; nursery, out-planting, and silvicultural stand management.
Fred M. van Eck Foundation for Purdue University: Funding for the van Eck Scholarships is provided by the Fred M. van Eck Foundation for Purdue University whose purpose is to support research in the genetic improvement of high-value North American hardwood tree species.
Qualifications: Candidates should have well developed quantitative skills and knowledge and experience in biological sciences such as forestry, horticulture, botany, plant biology, genetics, physiology, entomology, or pathology. Candidates should have a GPA of at least 3.4 and GRE scores averaging above the 60th percentile.
For fall semester, (August 2016), applications must be received by December 11, 2015 (M.S. and Ph.D).
For additional details visit the HTIRC website at http://www.htirc.org/
Have questions about tree planting? This series of publications can be viewed or downloaded free of charge. Planting and Care of Fine Hardwood Seedlings
van Eck Scholarships available for graduate research with the HTIRC.
Ask the HTIRC: email Lenny Farlee with your tree planting and forest management questions and we’ll help you find the answers.
